What a pharma omnichannel CoE is (and is not)
A pharma omnichannel Center of Excellence (CoE) is a small, cross-functional team that defines standards and decision rights so brands can execute consistently across channels. It exists to reduce variance, remove bottlenecks, and improve quality, measurement, and compliance readiness.
A CoE is not a “central team that builds every campaign.” In most organizations, the most resilient model is a hub-and-spoke approach: the CoE is the hub that sets guardrails, while brand pods and partners are the spokes that execute within those guardrails.
The compliance and data realities your CoE must design for
Your operating model has to work inside established regulatory and industry expectations for US prescription drug promotion. The core requirement to present information in a way that is not false or misleading is embedded in the FDA’s prescription drug advertising regulations under 21 CFR 202.1, which is why “speed” needs a governance system rather than heroics.
Omnichannel also amplifies privacy and consent complexity, especially when patient support programs, sign-ups, and digital identity are involved. If you touch protected health information, align your data handling and disclosures to the HIPAA Privacy Rule expectations and make those requirements operational in intake, tagging, access controls, and measurement design.
Finally, modern marketing operations increasingly relies on digital workflow evidence: approvals, versioning, and audit trails. When your process involves electronic records and electronic signatures for regulated activities, design controls that are compatible with 21 CFR Part 11 principles so your QA workflow is not just efficient, but defensible.
The omnichannel CoE operating model (who does what)
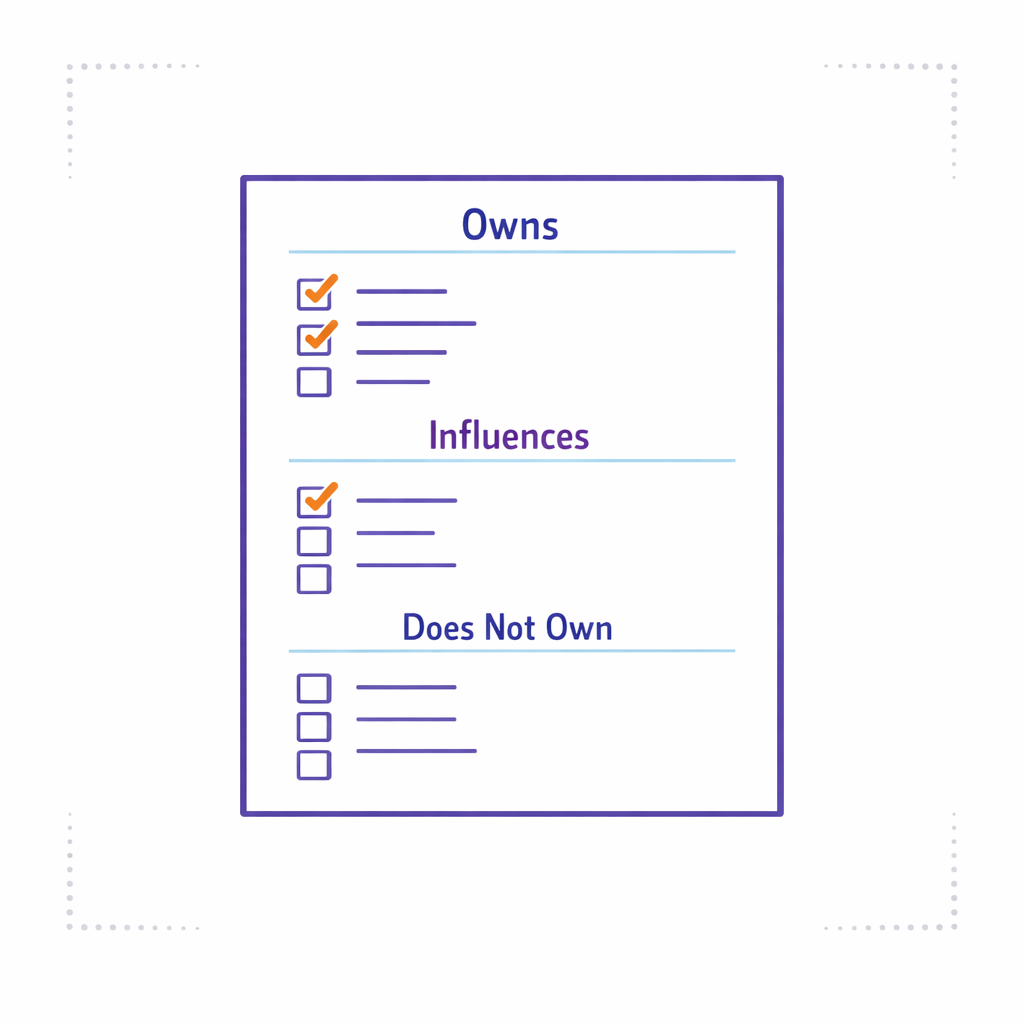
Start by writing a one-page charter that answers three questions: what the CoE owns, what it influences, and what it explicitly does not do. The goal is to reduce duplicate governance and clarify how decisions get made when teams disagree.
1) Define the CoE’s charter and decision rights
Common CoE decision rights that create leverage include: standard campaign intake, required metadata for content and journeys, minimum QA gates, analytics definitions, and the “approved tech stack” for orchestration and measurement. Keep the charter stable, then iterate the playbooks beneath it as tools and channels evolve.
2) Stand up a hub-and-spoke delivery structure
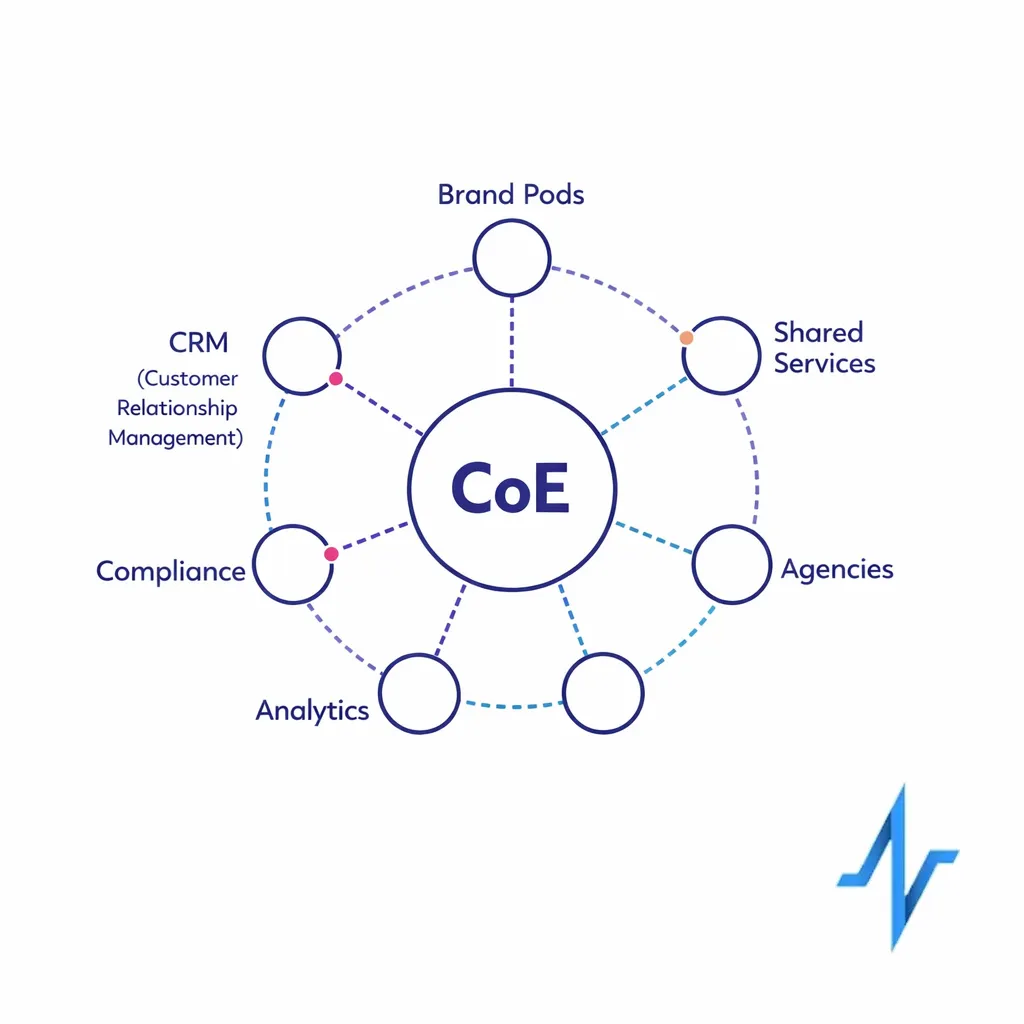
A practical structure is:
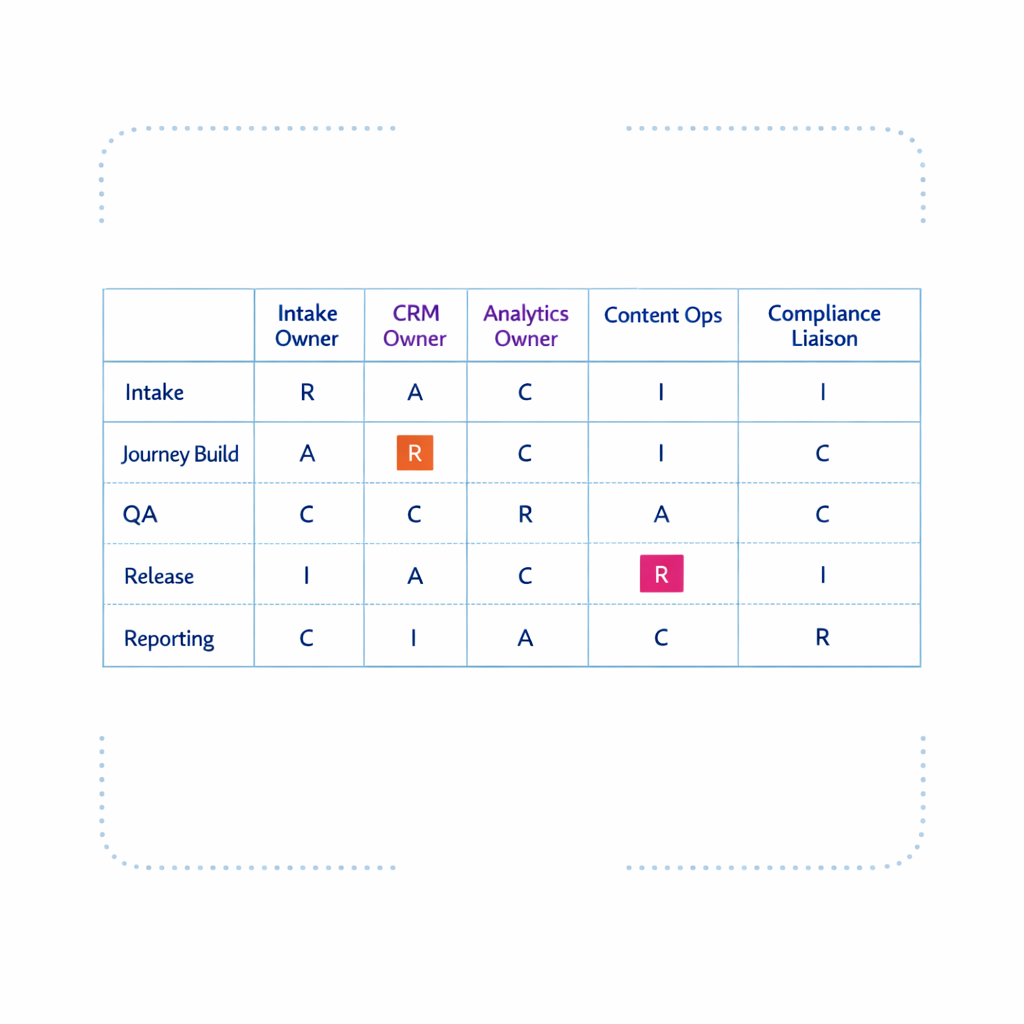
To keep the model lightweight, use a simple RACI and only formalize roles that drive throughput: intake owner, CRM owner, analytics owner, content ops lead, and compliance liaison.
- CoE (hub): standards, templates, training, measurement definitions, orchestration rules, and escalation paths.
- Brand pods (spokes): business strategy, creative direction, and channel execution for their indications and audiences.
- Shared services: MLR/PRC coordination, marketing ops, CRM operations, analytics, and martech enablement.
- Agencies/partners: build and run within the CoE’s SOPs, QA checks, and reporting requirements.
3) Establish a two-tier governance cadence
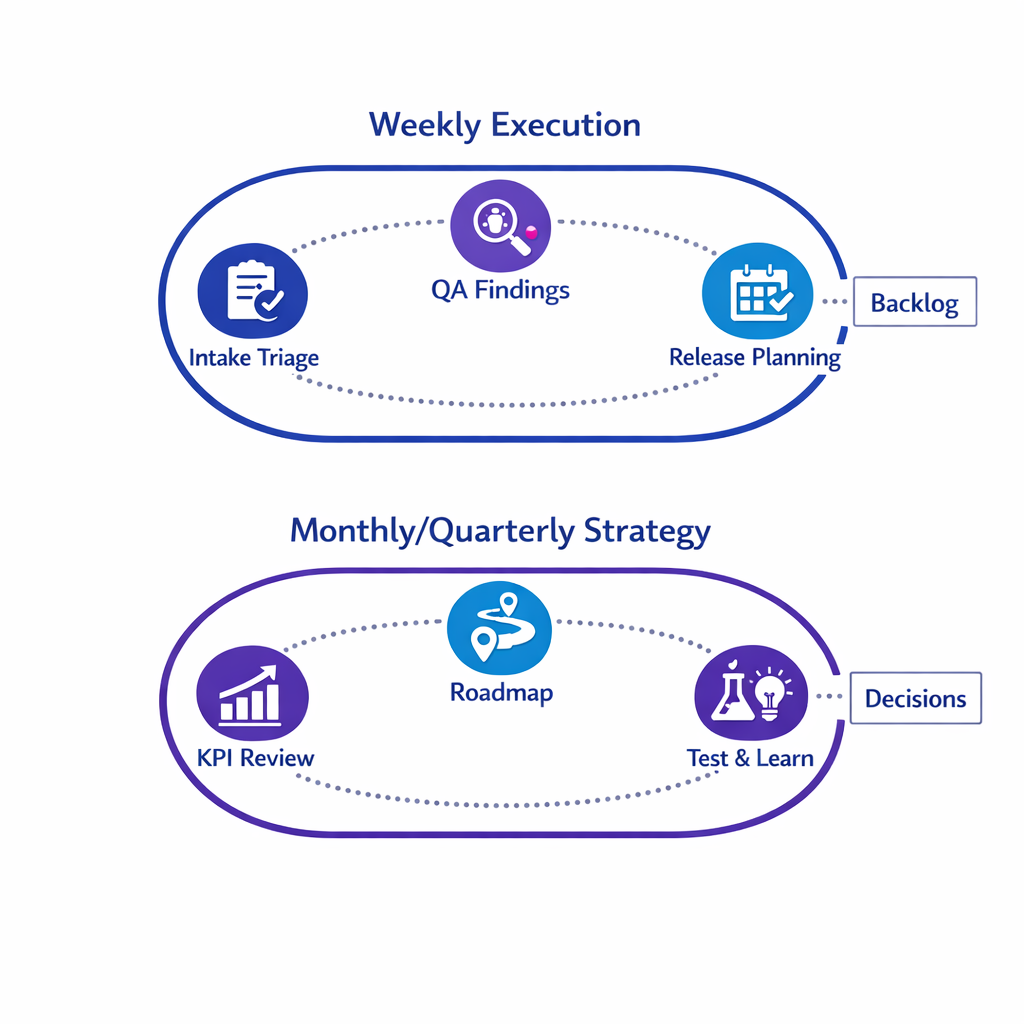
Most teams need two rhythms:
- Weekly execution governance: intake triage, in-flight issue resolution, QA findings, and release planning.
- Monthly/quarterly strategy governance: KPI review, channel performance, test-and-learn prioritization, and roadmap decisions.
Keep meeting outputs tangible: a prioritized backlog, documented decisions, and updated playbooks. If a meeting does not produce one of those artifacts, it is not governance, it is status.
The pharma governance framework (how work moves safely and predictably)
Pillar A: Campaign intake and prioritization
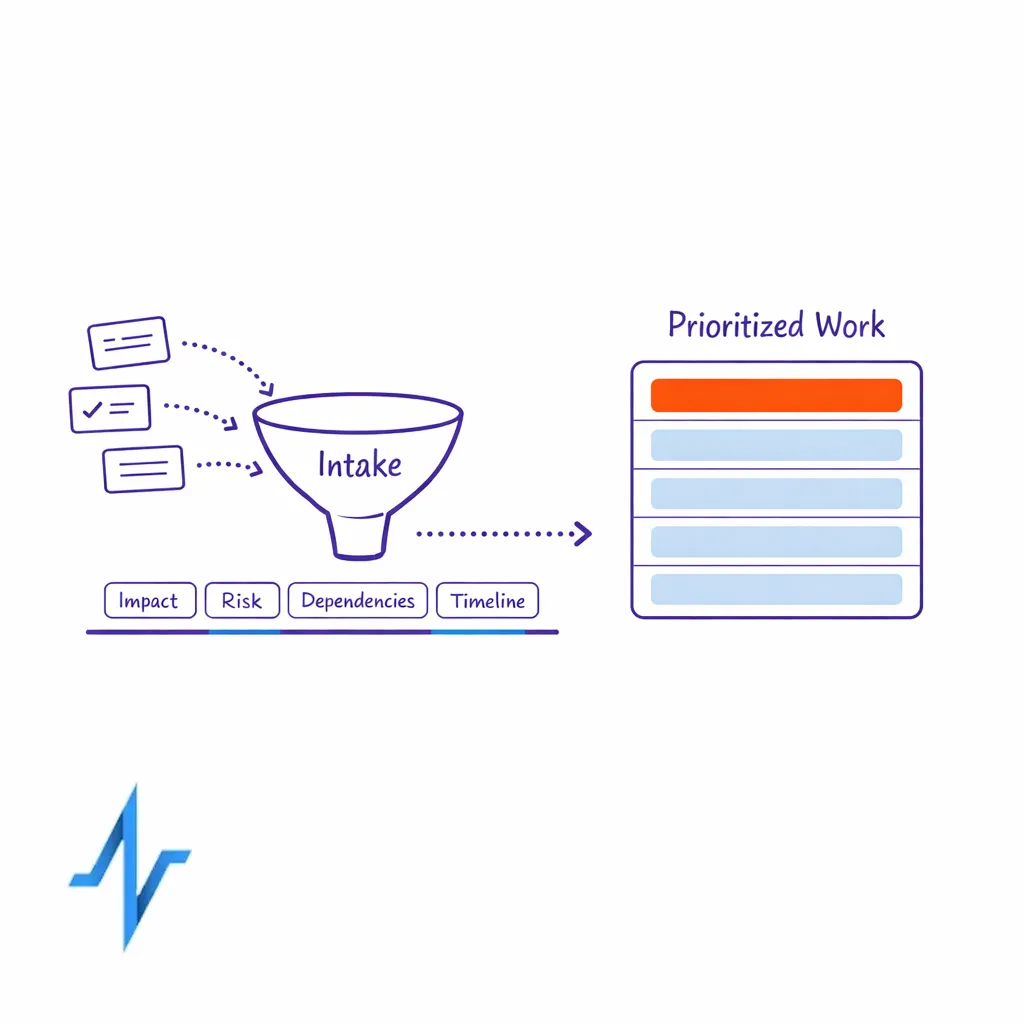
An effective campaign intake process is the foundation of scalable omnichannel execution. It prevents “side door” requests, makes capacity constraints visible, and creates a single place to capture compliance-relevant information early.
Design intake around a standardized brief that captures: audience (HCP vs patient/caregiver), objective, key claims and references, channel mix, data needs, measurement plan, and required review pathway. Add a lightweight scoring model so brands understand why certain work is prioritized (for example, patient impact, risk level, dependency on critical systems, and timeline constraints).
Pillar B: Modular content framework and content operations
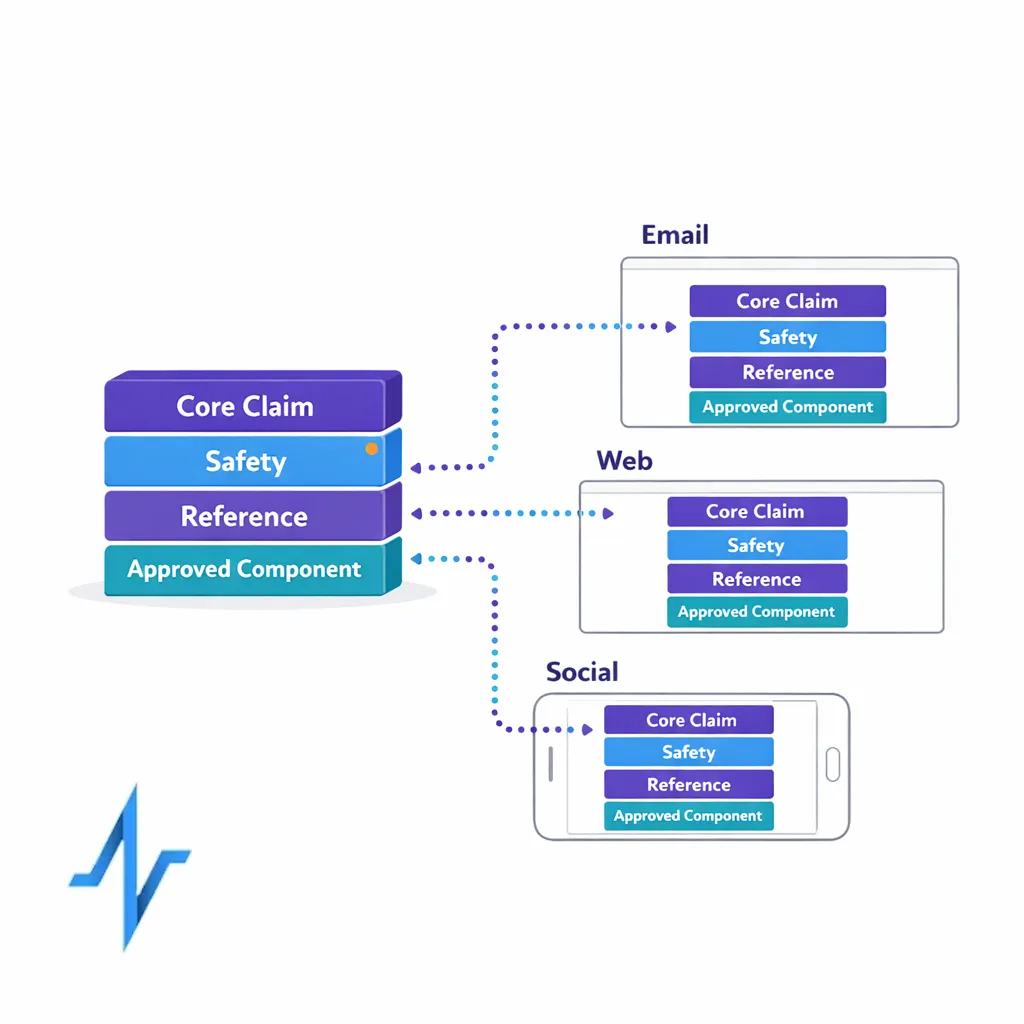
Omnichannel breaks when content is treated as one-off assets per channel. A modular content framework treats content as reusable building blocks: core claims and required safety language, approved components, and channel-specific wrappers.
Operationalize modularity with:
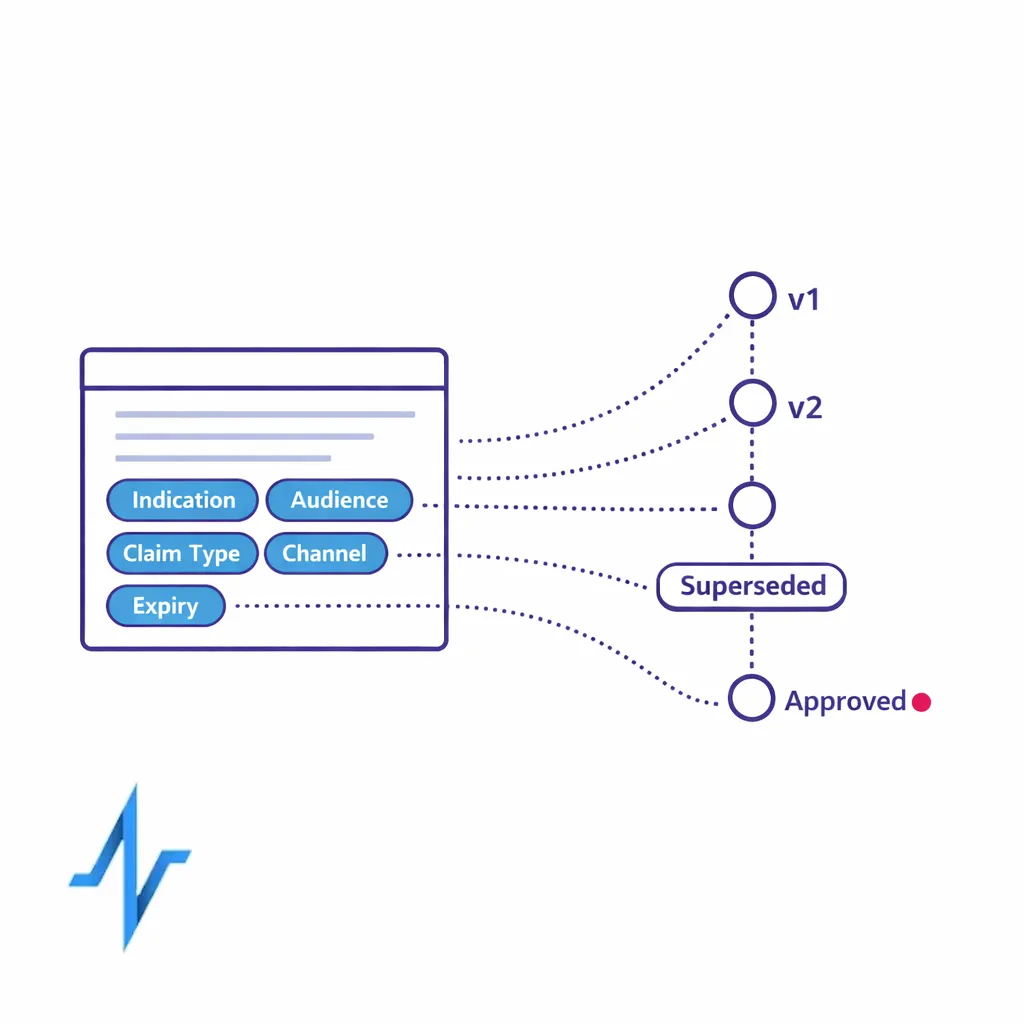
- Content metadata standards: indication, audience, claim type, references, channel eligibility, and expiration rules.
- Version control: a single source of truth for “what is approved” and what is superseded.
- Reuse rules: what can be repurposed, what requires re-review, and what cannot be adapted.
This is where a CoE can create significant efficiency. Even without promising specific time savings, reducing rework is a predictable outcome when teams stop rebuilding similar assets from scratch.
Pillar C: QA workflow and MLR/PRC readiness
Pharma omnichannel QA is not just “does the link work.” It includes content accuracy, risk presentation, targeting logic, suppression rules, and evidence that the right people approved the right version. Align your QA workflow to the way promotional materials are expected to be truthful and non-misleading under FDA prescription drug advertising requirements, and ensure your review approach fits the channel.
For digital and social channels, build playbooks that incorporate FDA’s thinking on online promotional communications described in FDA guidance on internet and social media platforms for prescription drug promotion. Even if you do not adopt every example, your governance should address the same practical issues: space constraints, link-outs, monitoring, and how risk information is presented alongside benefit information.
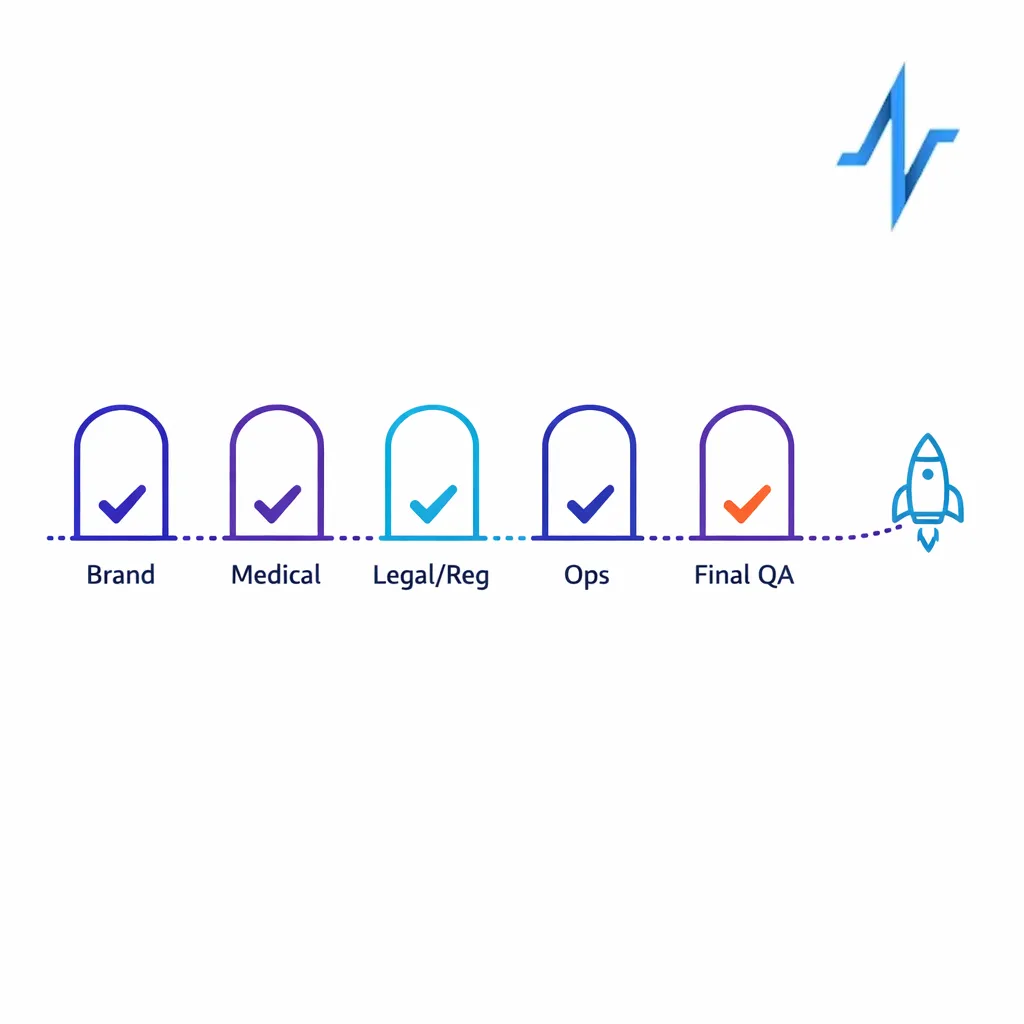
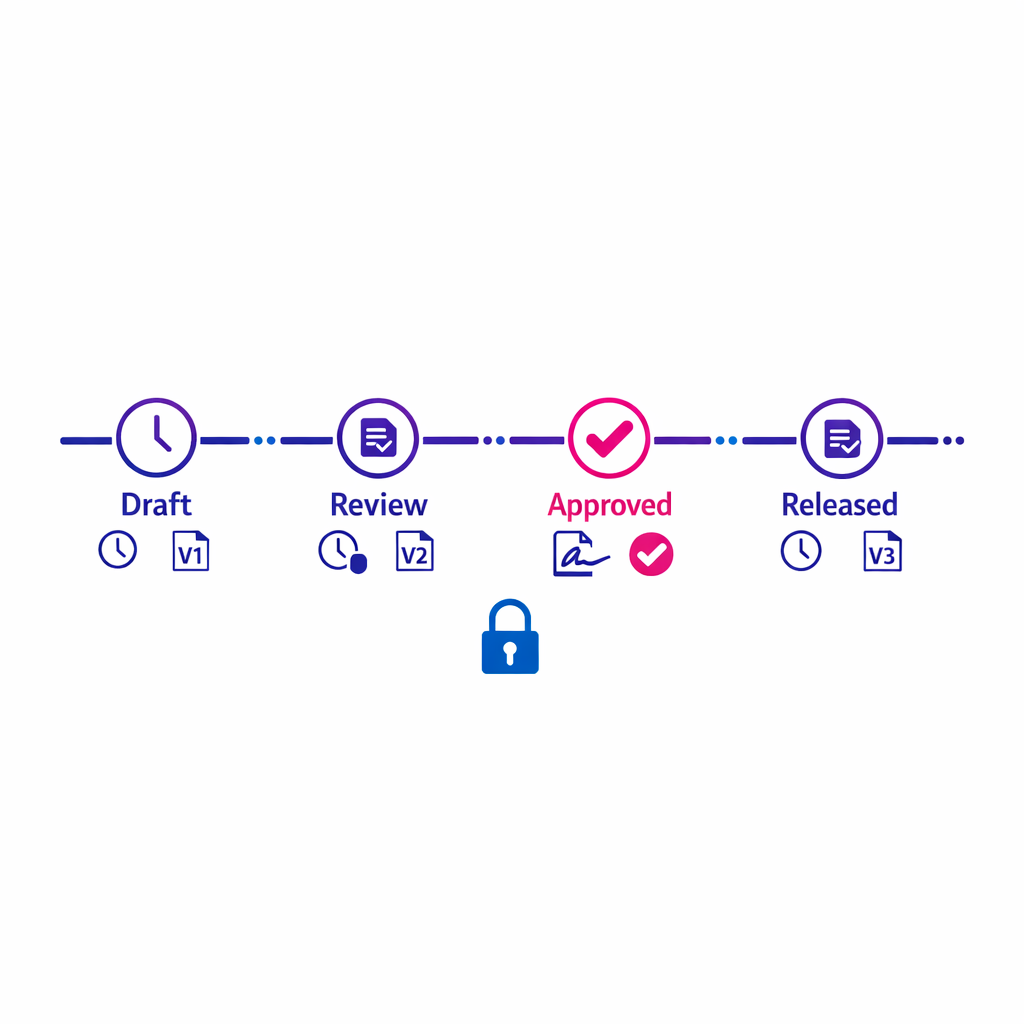
Finally, define what “release-ready” means. Many teams use a staged gate model: Brand sign-off, Medical sign-off, Legal/regulatory sign-off, Ops readiness (tracking, tagging, routing), and Final QA. Treat the checklist as a living SOP that evolves as your stack evolves.
Pillar D: CRM owner best practices and orchestration governance
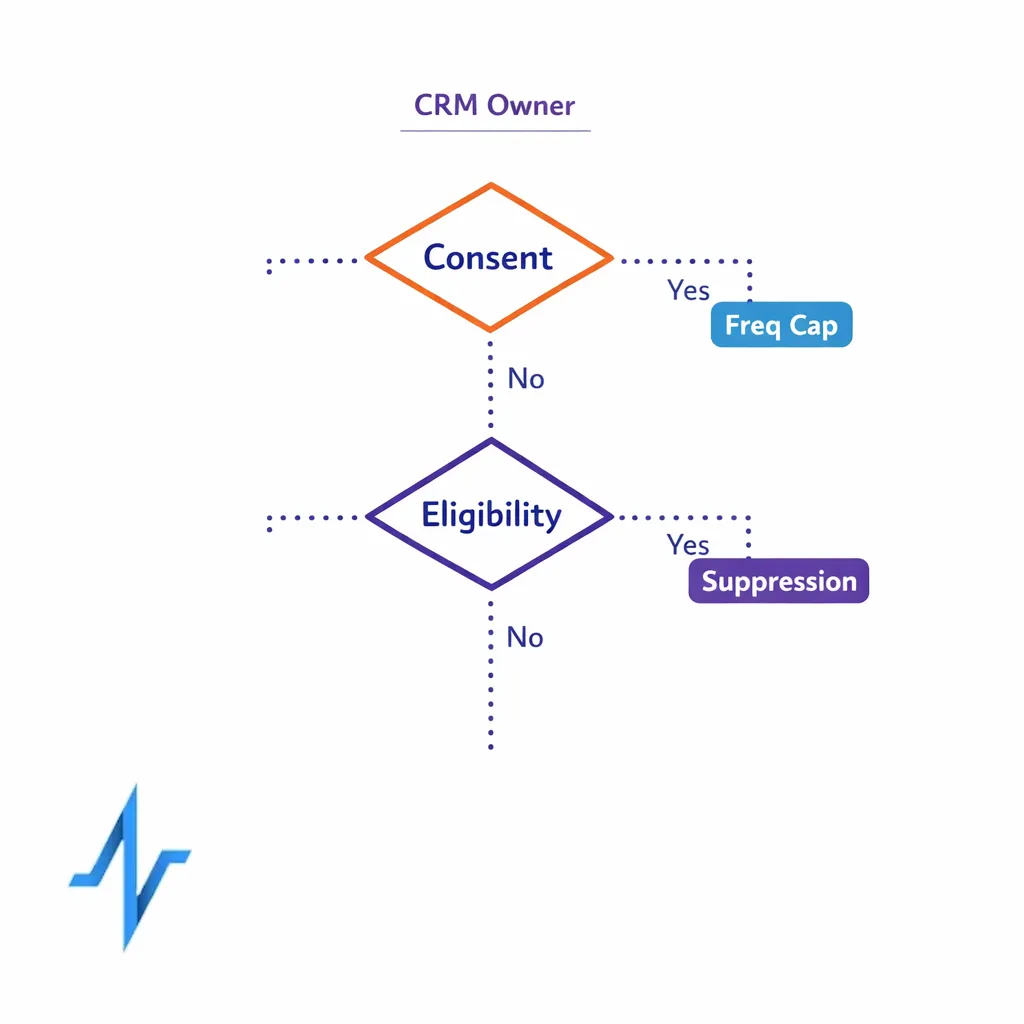
In pharma, the CRM owner is often the single most important role for scalable omnichannel execution. Their job is to keep the orchestration engine reliable: identity rules, HCP segmentation, consent flags, channel eligibility, frequency caps, and suppression lists.
Orchestration governance should answer:
- Who can change journey logic? Define who edits decision rules versus who requests changes.
- What is the escalation path? Create clear paths for field feedback, compliance concerns, and data quality issues.
- What are the non-negotiables? For example, honoring opt-outs, respecting channel preferences, and enforcing contact policies.
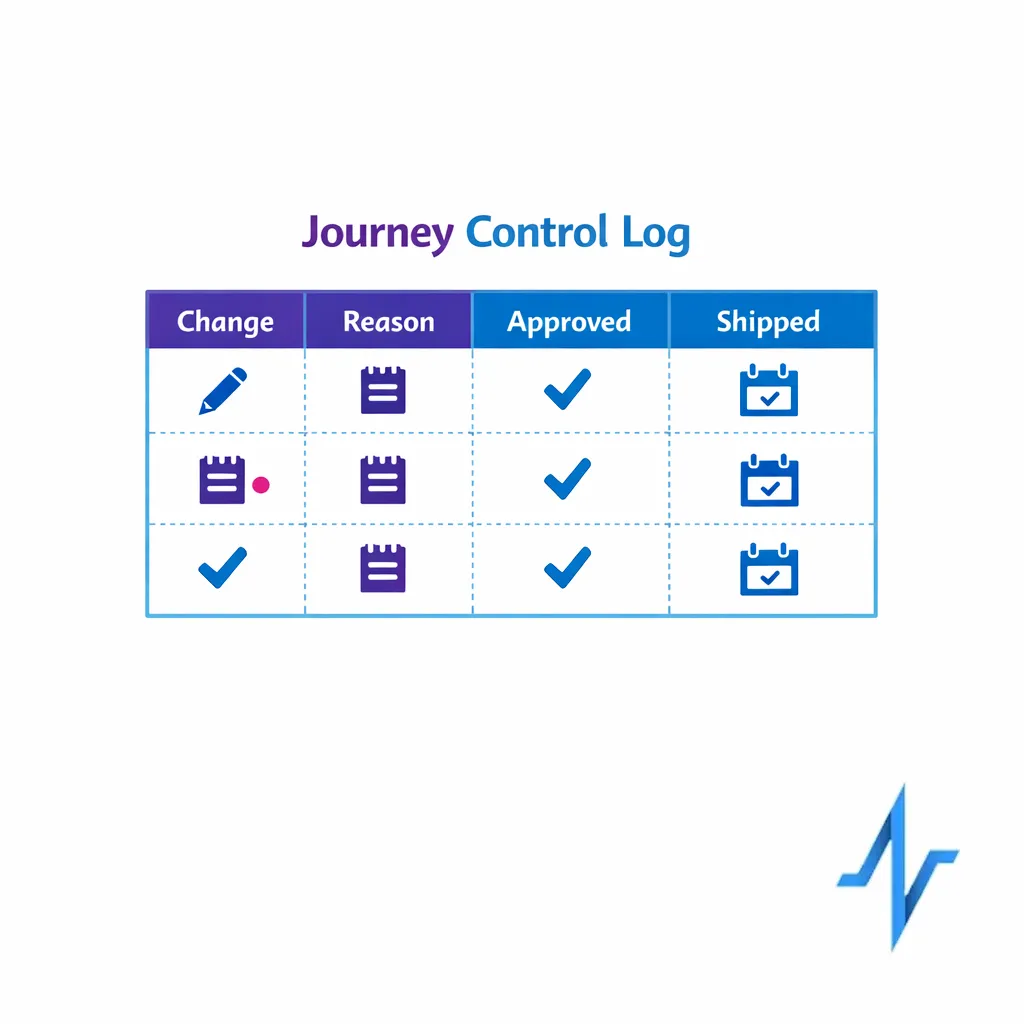
Keep a documented “journey control log” that records what changed, why it changed, who approved it, and when it shipped. This is both operationally useful and aligns with the expectation of maintaining reliable electronic records in regulated workflows, consistent with 21 CFR Part 11 principles where applicable.
Pillar E: Analytics governance and measurement design
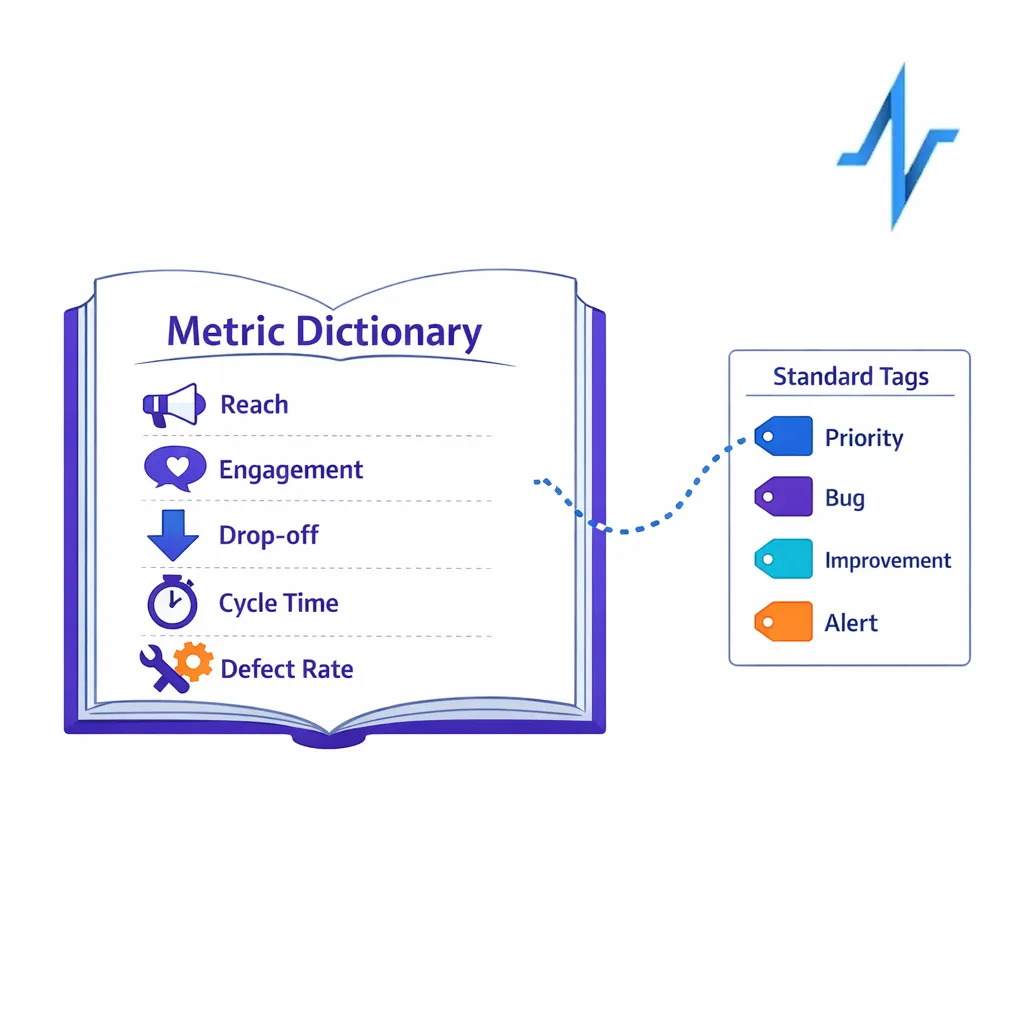
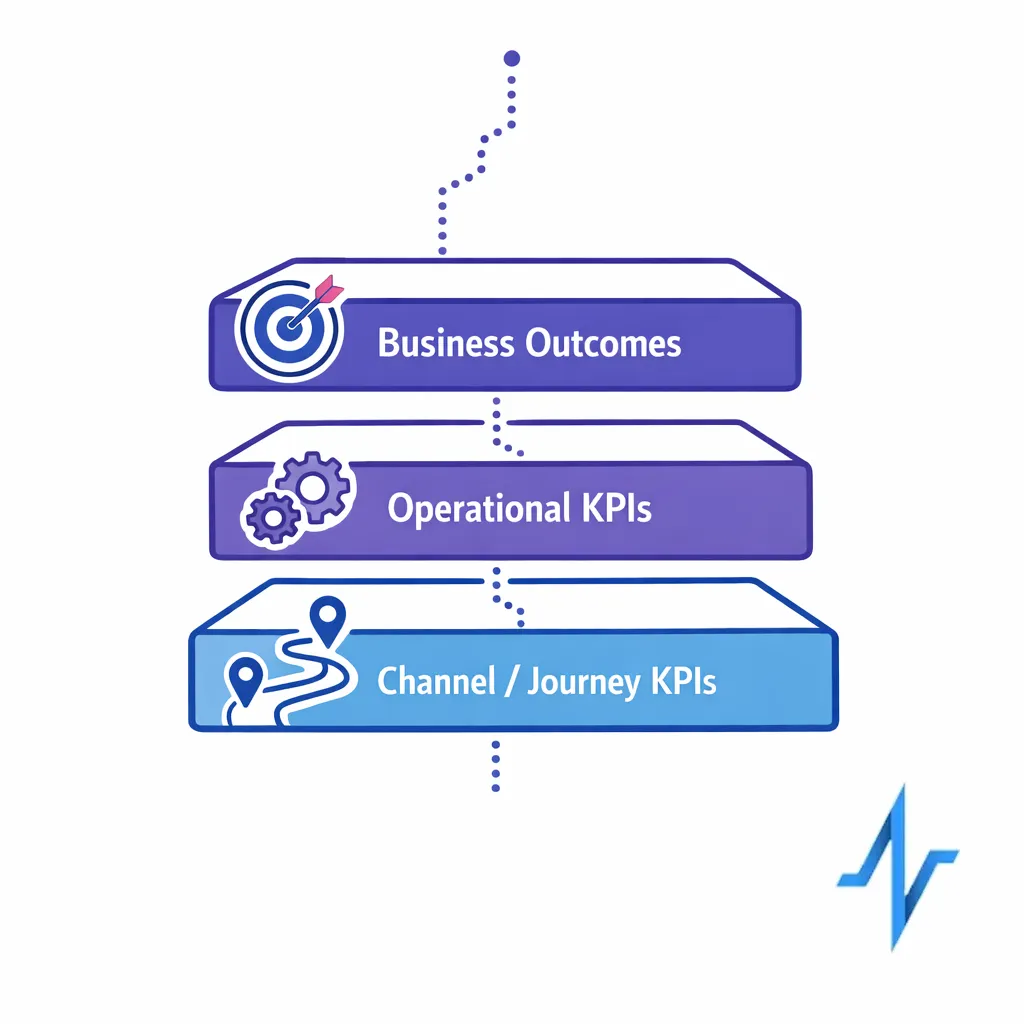
If every team defines success differently, an omnichannel program cannot scale. Analytics governance is the discipline of standardizing definitions (for example, what counts as an “engaged HCP”), ensuring instrumentation is consistent, and making performance visible in a way stakeholders trust.
Focus analytics governance on three layers:
- Business outcomes: what the brand is trying to change (education, adherence support, appropriate awareness) and how it will be evaluated.
- Operational KPIs: cycle time from intake to launch, QA defect rates, and rework rates.
- Channel/journey KPIs: reach, engagement, drop-off points, and handoffs between channels.
Where patient programs are involved, ensure measurement plans are privacy-aware and consistent with the handling expectations under the HIPAA Privacy Rule when PHI is in scope. Your CoE should be able to say, in plain language, what data is collected, why, and who can access it.
Pillar F: Partner governance (agencies, vendors, and affiliates)
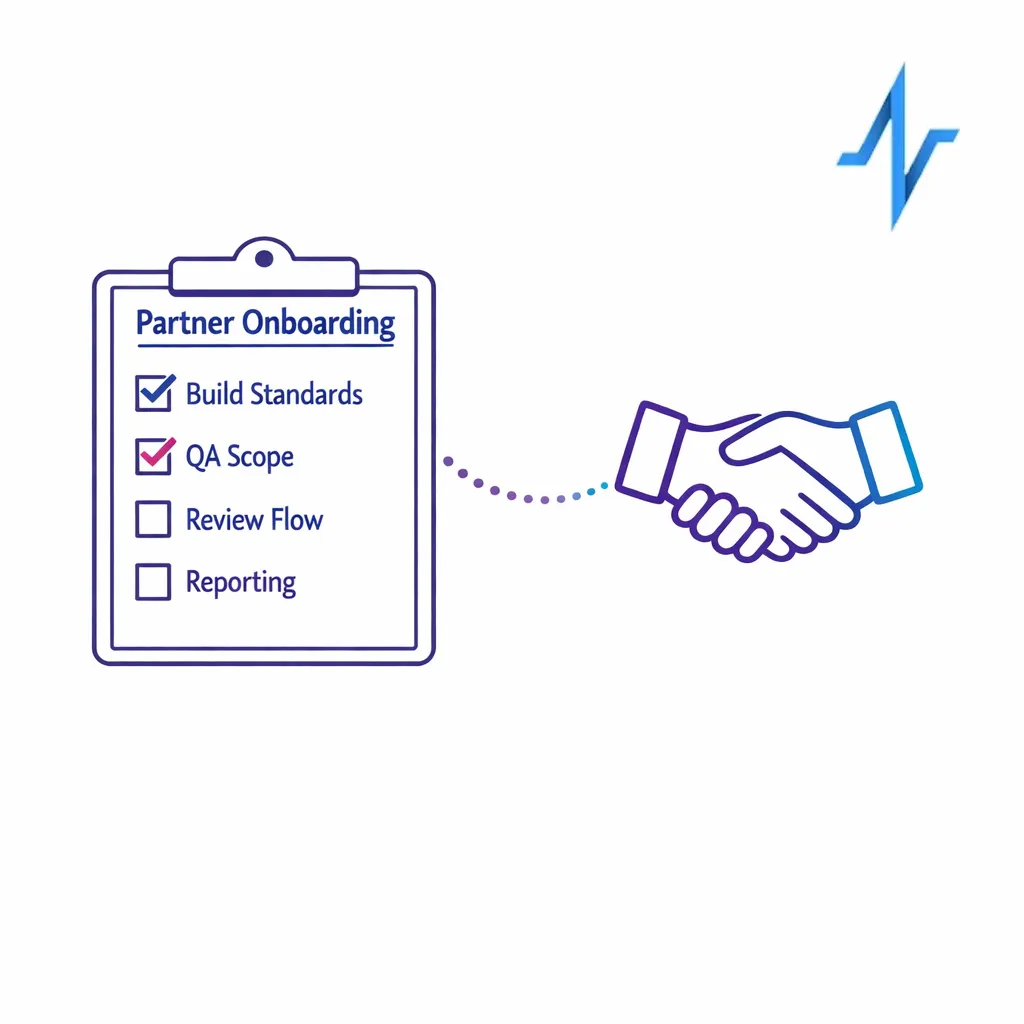
Most omnichannel programs rely on agencies and platform partners. Partner governance ensures outside teams can move quickly without introducing risk or creating unmaintainable one-off implementations.
Set clear expectations in SOWs and onboarding:
- Build standards: naming conventions, tagging, templates, and documentation requirements.
- QA responsibilities: what partners test versus what the internal team tests.
- Compliance responsibilities: how materials flow through review and what constitutes a material change.
- Reporting requirements: what must be delivered, how often, and in what format.
When aligning partner behavior to broader compliance programs, many organizations use government guidance as a reference point for “what good looks like,” including the HHS OIG’s Compliance Program Guidance for Pharmaceutical Manufacturers.
What changed (and what’s new) for omnichannel CoEs in 2026
Three shifts are pushing CoEs to become more operationally mature. First, omnichannel has moved from “more channels” to “connected decisions,” which means orchestration logic, identity, and measurement are now core assets that require governance, not ad hoc optimization.
Second, digital promotion expectations are clearer in practice, especially for online formats and platform constraints. Teams are increasingly expected to design processes that can support the realities covered in FDA’s internet and social media prescription drug promotion guidance, rather than treating social and digital as exceptions.
Third, enablement is now a differentiator. With frequent team changes and expanding partner ecosystems, the CoE that wins is the one that can onboard, certify, and monitor adherence to standards in a repeatable way, rather than relying on institutional memory.
Enablement plan: SOPs, templates, and training that actually stick
Step 1: Package the operating model into a CoE playbook
Make the model usable by turning it into a playbook with short, actionable SOPs. Each SOP should include purpose, scope, inputs/outputs, roles, required tools, and acceptance criteria.
Start with the high-impact SOPs:
- Campaign intake process pharma: request form, required fields, triage rules, and SLAs.
- Standard operating procedures omnichannel: how journeys are built, tested, approved, and released.
- QA workflow pharma marketing: test plan templates, defect severity definitions, and sign-off requirements.
- Analytics governance pharma: metric dictionary, tagging standards, and reporting cadence.
Step 2: Build a reusable asset kit (templates and “golden paths”)
Templates reduce variability and speed up delivery, especially when multiple agencies are building in parallel. Keep templates channel-agnostic where possible and provide “golden path” examples that show the approved way to build common journeys.
Useful templates include: campaign brief, modular content matrix, journey logic worksheet, UTM and event tagging guide, QA checklist, and a release note format. When these are standardized, it becomes easier to compare performance across brands and reduce reinvention.
Step 3: Launch a training program with role-based tracks
Training should map to the roles in your operating model, not to organizational charts. A CRM owner track should cover orchestration governance, identity rules, and operational monitoring. A brand marketer track should cover intake, content modularity, and how decisions impact measurement.
If you want training to become a scalable capability, turn it into a certification. Many teams formalize this with an internal badge (for example, “Pulse Certified”) tied to hands-on proficiency, not attendance.
Step 4: Instrument adoption and quality, not just campaign performance
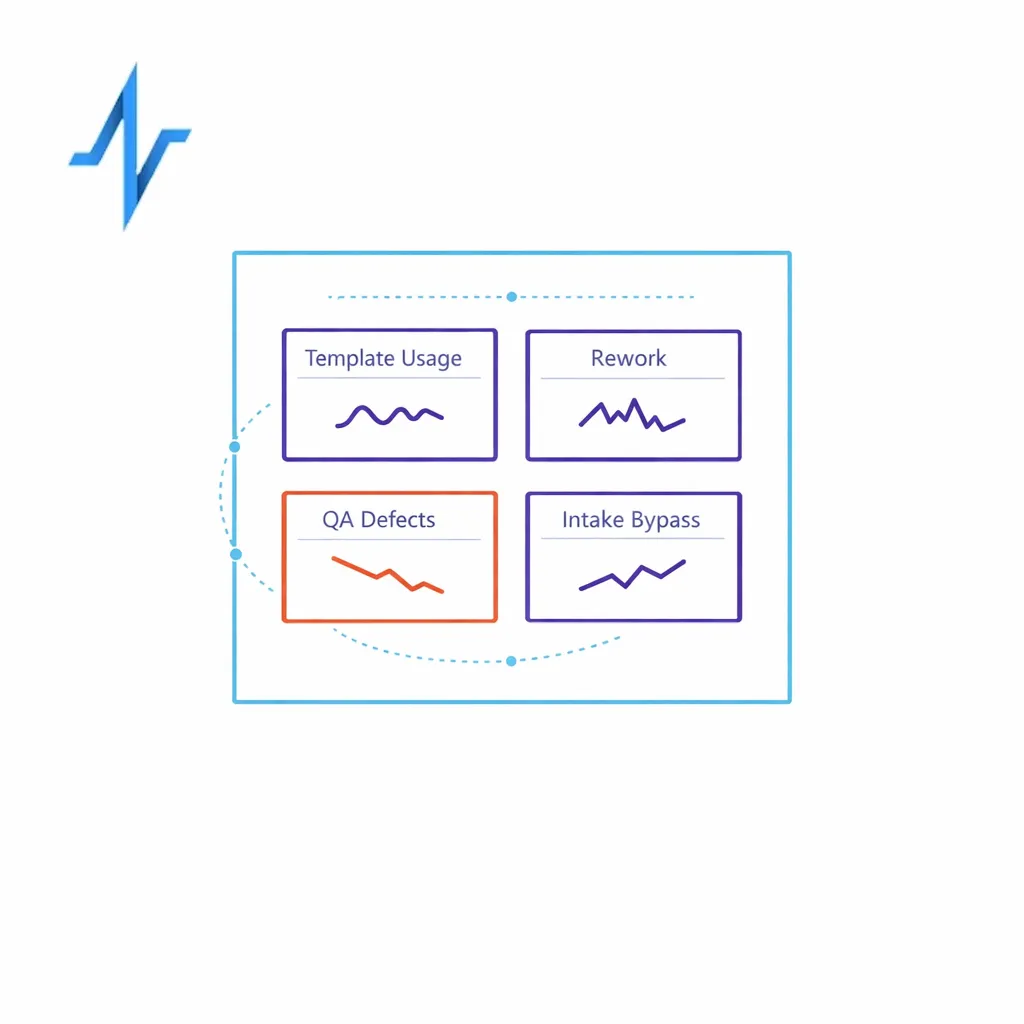
CoEs often measure campaign outcomes but fail to measure whether the operating model is being followed. Track leading indicators like template usage, rework rates, QA defect trends, and how often teams bypass intake.
Use those signals to update enablement content. If the same issue recurs, that is not an individual problem; it is a missing standard, a confusing SOP, or a workflow gap.
Common mistakes and misconceptions
Mistake 1: Treating governance as “more meetings”
Governance is decision-making with documentation. If your governance does not change what gets built, how it gets built, or how it is measured, you are adding overhead without control.
Mistake 2: Building omnichannel around channels instead of journeys
Channel teams optimize what they own. Without journey-level orchestration governance, you end up with duplicated touches, inconsistent sequencing, and measurement that cannot answer cross-channel questions.
Mistake 3: Leaving compliance and privacy requirements to the end
Late-stage review creates fire drills and encourages “minimize changes” behavior, which can lock in weak measurement designs and brittle implementations. Bringing compliance and privacy considerations into intake and modular content design reduces downstream churn and supports expectations around truthful, non-misleading promotion under FDA advertising requirements.
Mistake 4: Treating analytics as a reporting task instead of a governance task
Dashboards cannot fix inconsistent tagging, unclear definitions, or missing identity rules. Analytics governance needs owners, standards, and enforcement, just like content and orchestration do.
What to do next: a practical CoE launch checklist
- Write the CoE charter: scope, decision rights, and escalation paths.
- Publish a simple RACI: intake owner, CRM owner, analytics owner, content ops lead, and compliance liaison.
- Stand up intake: one request path, triage rules, and prioritization criteria.
- Define release readiness: QA gates, sign-offs, and documentation requirements aligned to your regulated workflow needs (including 21 CFR Part 11 considerations where applicable).
- Standardize modular content: metadata, versioning, reuse rules, and a single source of truth.
- Lock measurement foundations: metric dictionary, tagging standards, and reporting cadence.
- Codify orchestration governance: journey change controls, frequency caps, consent handling, and suppression rules.
- Onboard partners: build standards, QA responsibilities, and reporting requirements in SOWs.
- Launch role-based training: provide job-specific tracks and a certification path (for example, Pulse Certified) to make skills portable across teams.
See how Pulse Health can support a scalable omnichannel CoE
If your goal is scalable omnichannel execution with consistent governance, Pulse Health can help you operationalize the CoE playbook in day-to-day work: standard intake, repeatable orchestration patterns, QA-ready workflows, and measurement definitions that stay consistent across brands and partners.
To evaluate fit for your operating model, you can Request a Demo, Book a Consultation, or Talk to Pulse Health. If you are mapping martech dependencies, you can also Get the Platform Overview before you finalize your CoE roadmap.


