Pharma omnichannel teams are under pressure to prove what worked across HCP engagement and patient programs, without increasing privacy risk or creating reporting chaos across agencies and data partners. The hard part is not collecting “more data.” It is building a measurement architecture that can align identities and events across channels while staying de-identified, purpose-limited, and operationally scalable.
This article is for US pharma brand teams and omnichannel leads, commercial ops and CRM owners, and the agencies and partners who deliver media, sites, and patient education flows. You will learn what a de-identified measurement layer is, how tokenization and identity resolution fit together, how to align partner data into a consistent omnichannel reporting framework, and what to implement next.
Why measurement breaks in real-world pharma omnichannel
Most “cross-channel attribution pharma” efforts fail for the same reason: each partner brings its own identifiers, taxonomy, and reporting logic. One dashboard counts impressions, another counts “engaged sessions,” and your CRM counts leads, but none of it joins cleanly into a single de-identified measurement pharma view.
At the same time, privacy-by-design analytics is no longer optional. Even when the business goal is legitimate (program optimization, budget allocation, patient education), measurement must be built so sensitive data is minimized, access-controlled, and separated from activation whenever possible.
What a de-identified measurement layer is (and what it is not)
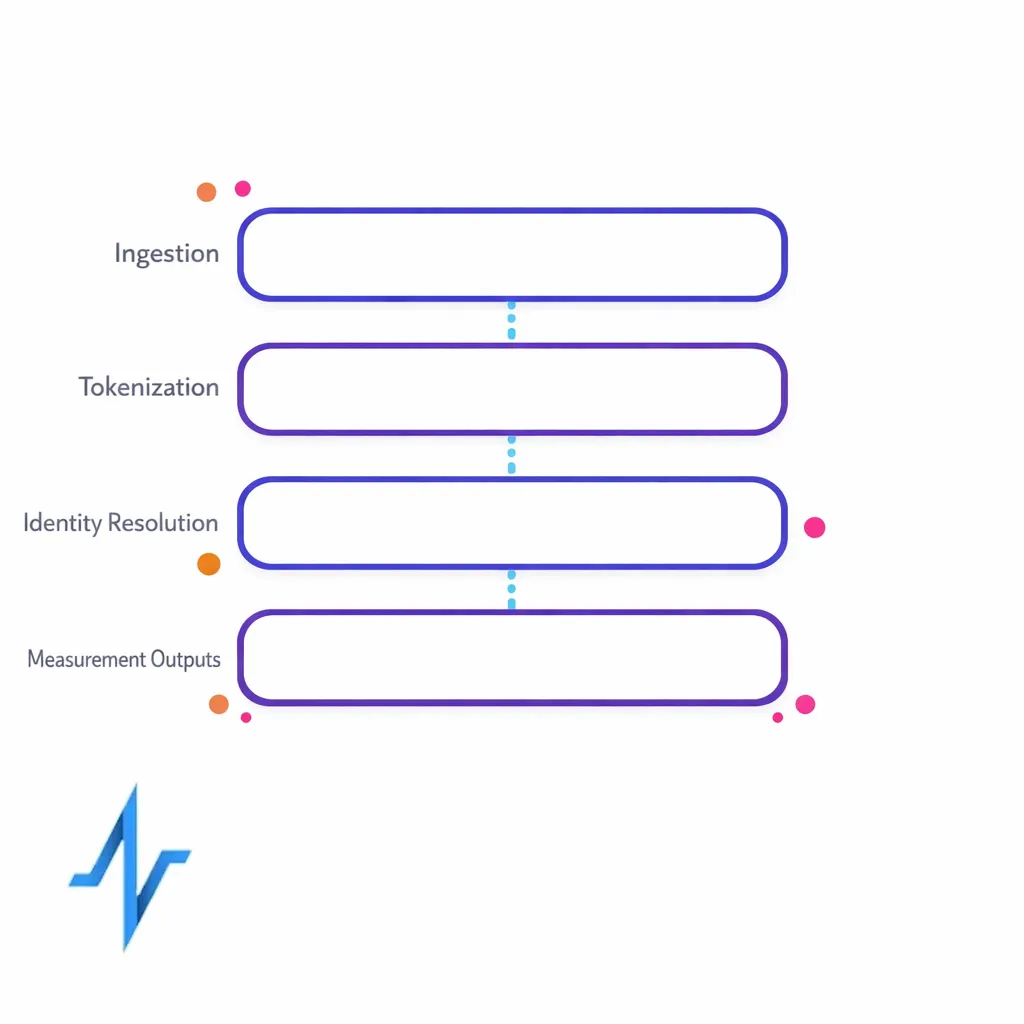
A de-identified measurement layer is the middle tier between execution systems (sites, email, media partners, call centers, hubs) and analytics outputs (dashboards, MMM, lift studies). It standardizes events, transforms identifiers into privacy-preserving tokens, resolves identities at the right level of granularity, and produces measurement-ready datasets that partners can align to consistently.
It is not a single vendor report, a one-off “identity graph healthcare marketing” project, or a spreadsheet stitch-up at the end of the quarter. It is measurement architecture pharma teams can run continuously, with governed inputs and repeatable logic.
Compliance foundations: de-identification, tokenization, and “don’t mix purposes”
Start by anchoring terminology. Under HHS guidance on de-identification under the HIPAA Privacy Rule, “de-identified” has a specific meaning and is not interchangeable with “pseudonymous” or “hashed.”
HIPAA provides two primary approaches for de-identification, the Safe Harbor method and the Expert Determination method in 45 CFR 164.514. In measurement architecture terms, tokenization is a technique you may use inside a broader de-identification strategy, but tokenization alone does not automatically make data de-identified.
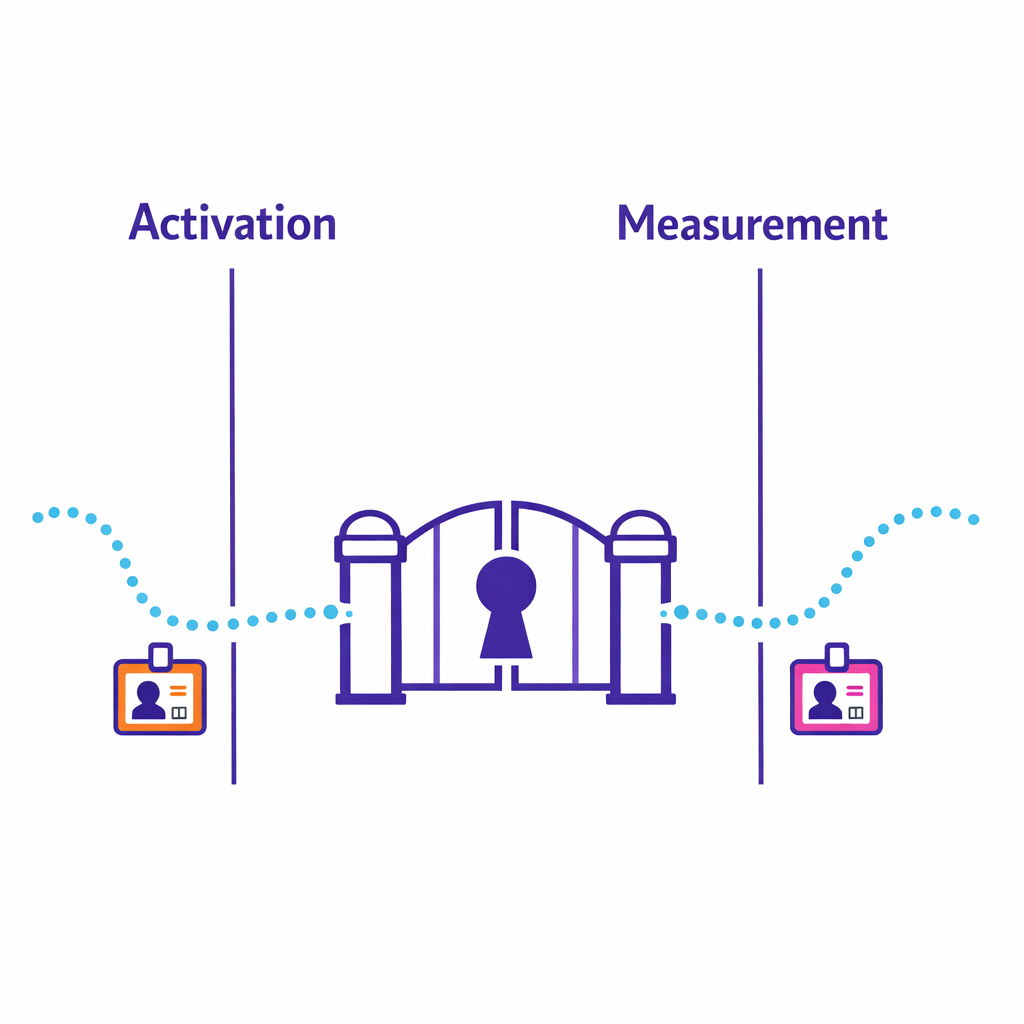
For brand and ops teams, the most practical compliance-aware design principle is separation of concerns. Build the measurement layer so that: (1) it collects only what is needed for defined use cases, (2) it limits direct identifiers, (3) it restricts who can access any lookup tables or keys, and (4) it avoids using measurement datasets for activation unless you have a clear, permitted workflow and governance.
The reference architecture: four layers that stay stable even as channels change
Channels and vendors will rotate. Your measurement layer should not. A durable omnichannel reporting framework for HCP and patient programs typically includes four layers.
- Ingestion and normalization: Collect events from web, email, CRM, hubs, call centers, and partners into a standard schema.
- Tokenization and privacy transforms: Convert identifiers into consistent, purpose-bound tokens and remove or quarantine direct identifiers.
- Identity resolution: Perform HCP identity matching and patient identity resolution at the appropriate level (person, household, account, or program enrollment).
- Measurement outputs: Produce de-identified datasets for reach/frequency, journeys, conversion, lift, and partner performance alignment.
If you design around these layers, “cookieless identity solutions” and channel policy shifts become implementation details rather than existential threats to your reporting.
Tokenization for healthcare marketing: making identifiers align without exposing identities
Tokenization healthcare marketing workflows exist to solve a practical problem: partners need a join key to align events, but you want to avoid sharing direct identifiers. The right approach depends on your governance model, partner ecosystem, and whether you need deterministic matching across sources.
Deterministic tokens (useful for partner alignment)
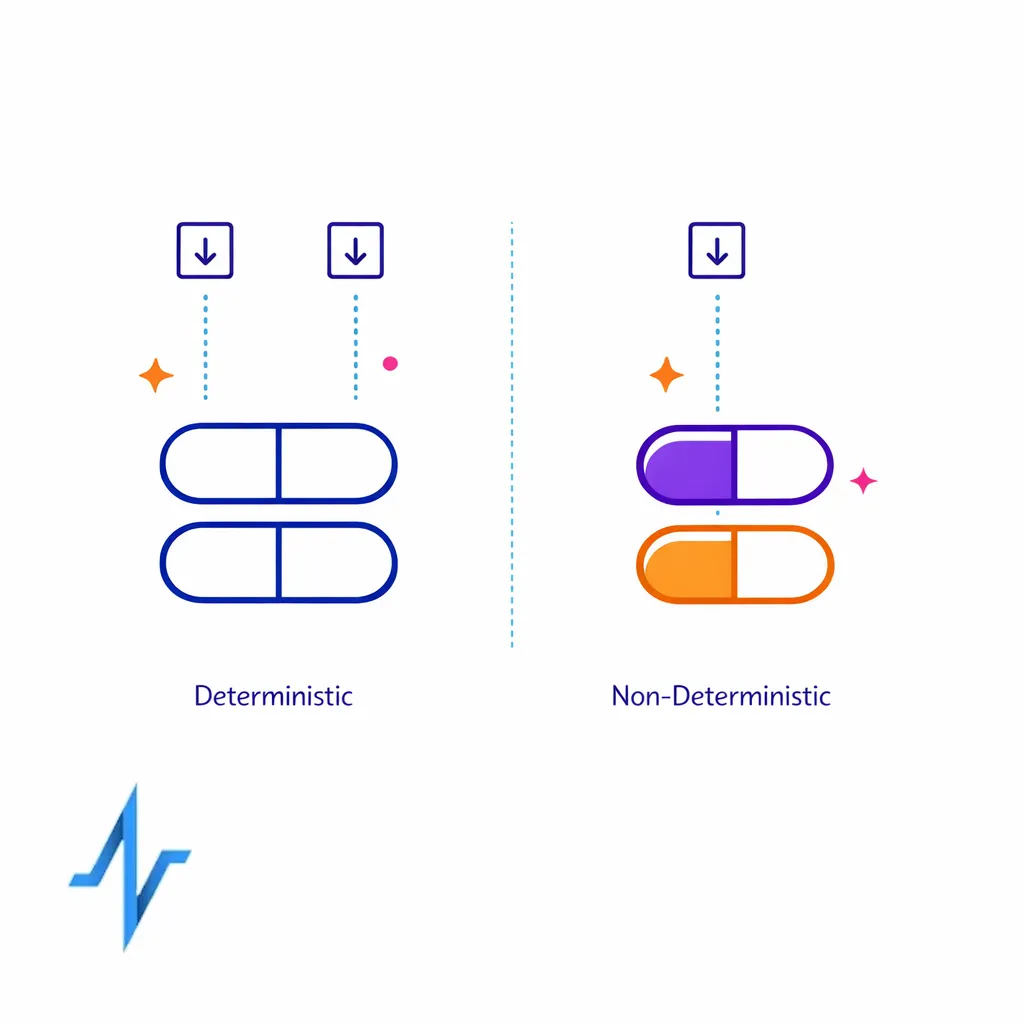
Deterministic tokenization produces the same token for the same input under a controlled method. In plain terms, if two partners see the same email address (or another permitted input), they can generate the same hashed identifiers pharma teams can match on, as long as both use the same algorithm and normalization rules.
Because deterministic tokens enable joining, they require stronger controls. Treat the tokenization method as part of your measurement infrastructure, not a casual “hash it and ship it” step. Define normalization (case, whitespace, phone formatting), decide whether tokens are program-specific, and restrict who can generate tokens and where.
Non-deterministic tokens (useful for internal privacy boundaries)
Non-deterministic tokenization (or random tokens) produces different outputs even for the same input. This is often useful when you want to measure within a system without enabling broad cross-partner matching. It can reduce linkage risk but typically requires an internal mapping service to reconcile events.
Many pharma teams end up with a hybrid model: deterministic tokens for carefully governed partner data alignment, and non-deterministic tokens for internal segmentation boundaries or system-to-system routing.
Design choices that prevent “measurement keys” from becoming activation IDs
One of the most common governance failures is that a measurement join key slowly turns into a universal ID used for everything. You can reduce that risk through architectural controls.
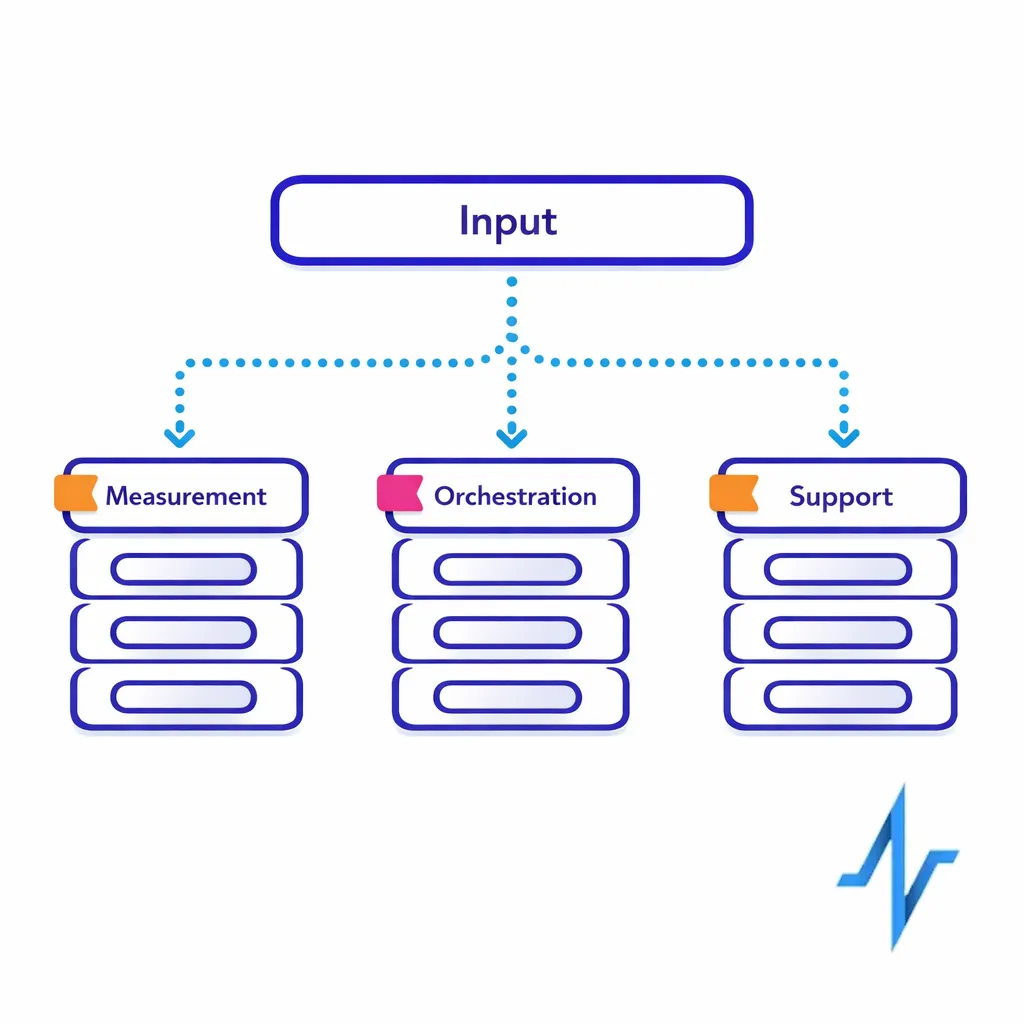
- Purpose-bound token namespaces: Use different tokens for measurement vs orchestration vs support operations, even if they derive from the same raw input.
- Rotation and versioning: Build for method changes without breaking historical reporting by tracking token versions.
- Minimize fields required for matching: Use the least sensitive inputs that still meet match-rate and accuracy needs.
This is the core of consent-based measurement in practice: measurement that is technically capable of joining what must be joined, while remaining constrained by design.
Identity resolution pharma teams can actually operate
Identity resolution is where measurement architecture usually becomes fragile. The fix is to define “identity” per use case, not per vendor. HCP identity matching may need person-level resolution, while patient program measurement might need an enrollment-level or household-level view.
HCP identity matching: start with NPI, but do not stop there
For US HCP programs, NPI-based identity is a natural anchor for measurement. The National Provider Identifier (NPI) standard described by CMS exists to support consistent identification of health care providers in standard transactions, and it can be a stable backbone for cross-channel reporting when partners can supply NPI or when you can enrich to it through governed workflows.
However, “NPI solves identity” is a misconception. In practice, HCP identity resolution often needs to account for:
- Multiple practice locations and affiliations: Engagement may happen at the HCP level, while outcomes you can observe may be at the account or facility level.
- Role and specialty drift: Identity is stable, but targeting and relevance depend on up-to-date attributes.
- Partner match variability: Some channels provide NPI directly; others require matching via intermediate keys.
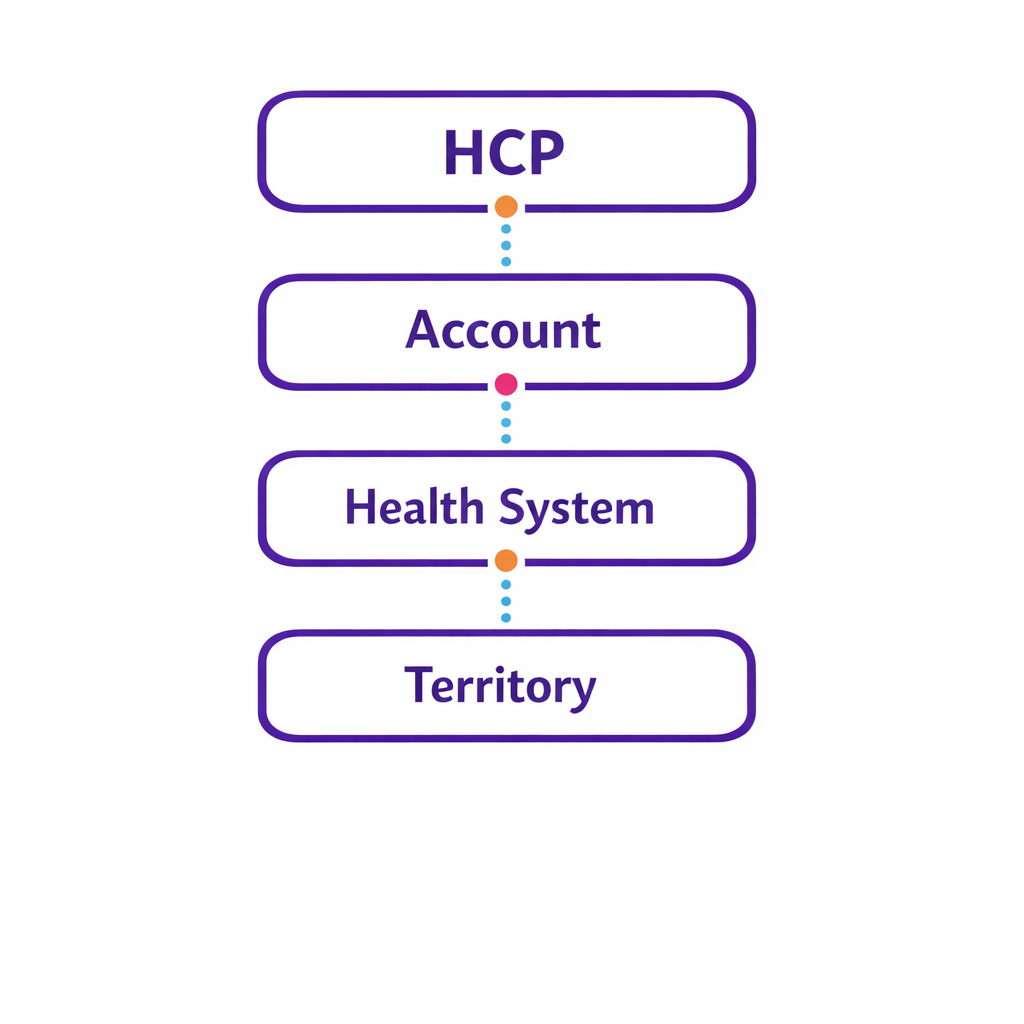
A strong measurement layer treats NPI as one graph node, not the entire graph. You should be able to report at multiple levels: HCP, account, health system, and territory, without rewriting your entire pipeline every time a data partner changes.
Patient identity resolution: enrollment-first, privacy-first
Patient education and support programs introduce a different identity problem. You often need to measure acquisition sources (which channels drove sign-ups), engagement (education content completion, refill reminders), and persistence over time.
In a de-identified measurement layer, patient identity resolution typically centers on a program enrollment identifier, then links downstream events to that enrollment using tokens. This supports patient program measurement without forcing every system to handle direct identifiers, and it allows you to separate operational workflows (support, fulfillment) from analytics outputs.
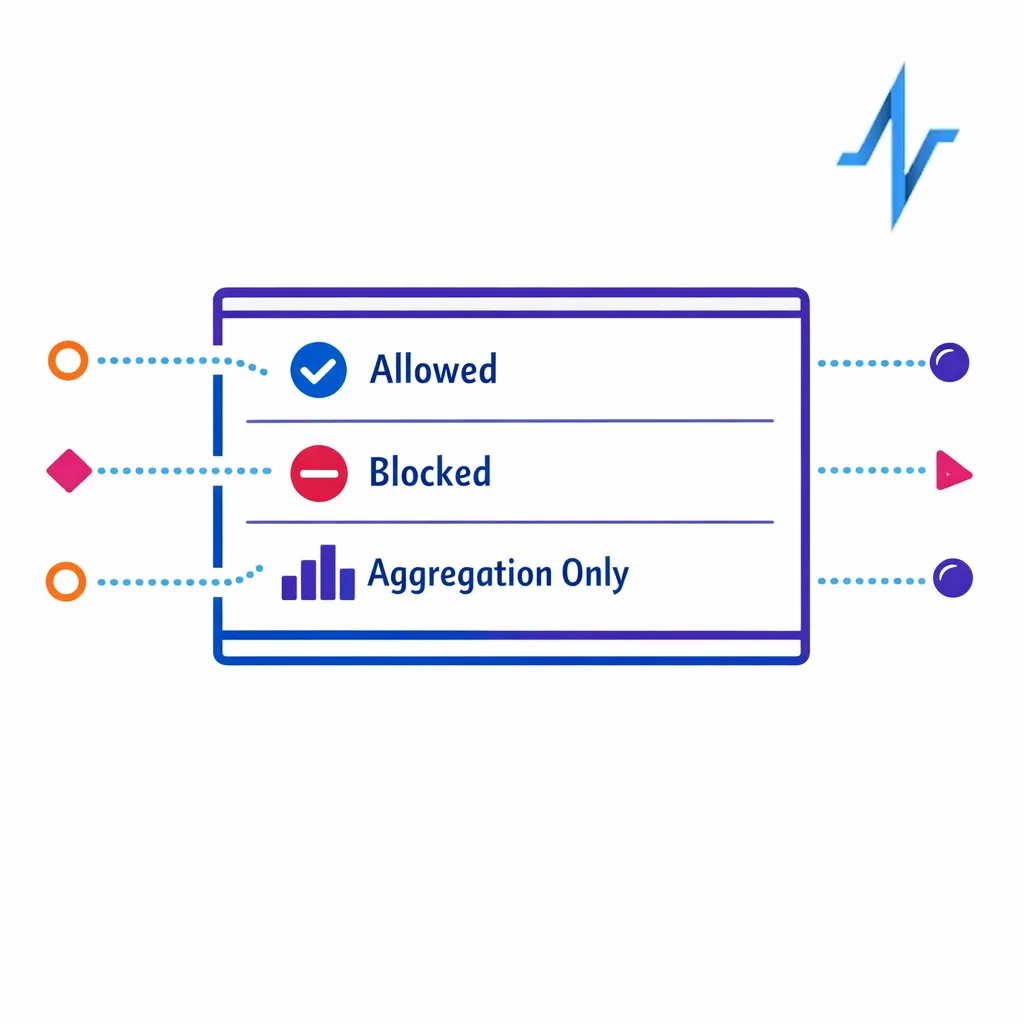
Consent-based measurement becomes practical when you explicitly track what the user agreed to and what the program stated it would do, then constrain which measurement joins you allow. Operationally, this shows up as rules in your pipeline: allowed joins, blocked joins, and “aggregation only” outputs for certain sources.
Partner data alignment: the unglamorous work that makes reporting trustworthy
Most omnichannel reporting issues are not “analytics problems.” They are partner alignment problems. If agencies, publishers, hubs, and CRM systems do not share a consistent event taxonomy and identity approach, you will spend every quarter debating definitions instead of optimizing performance.
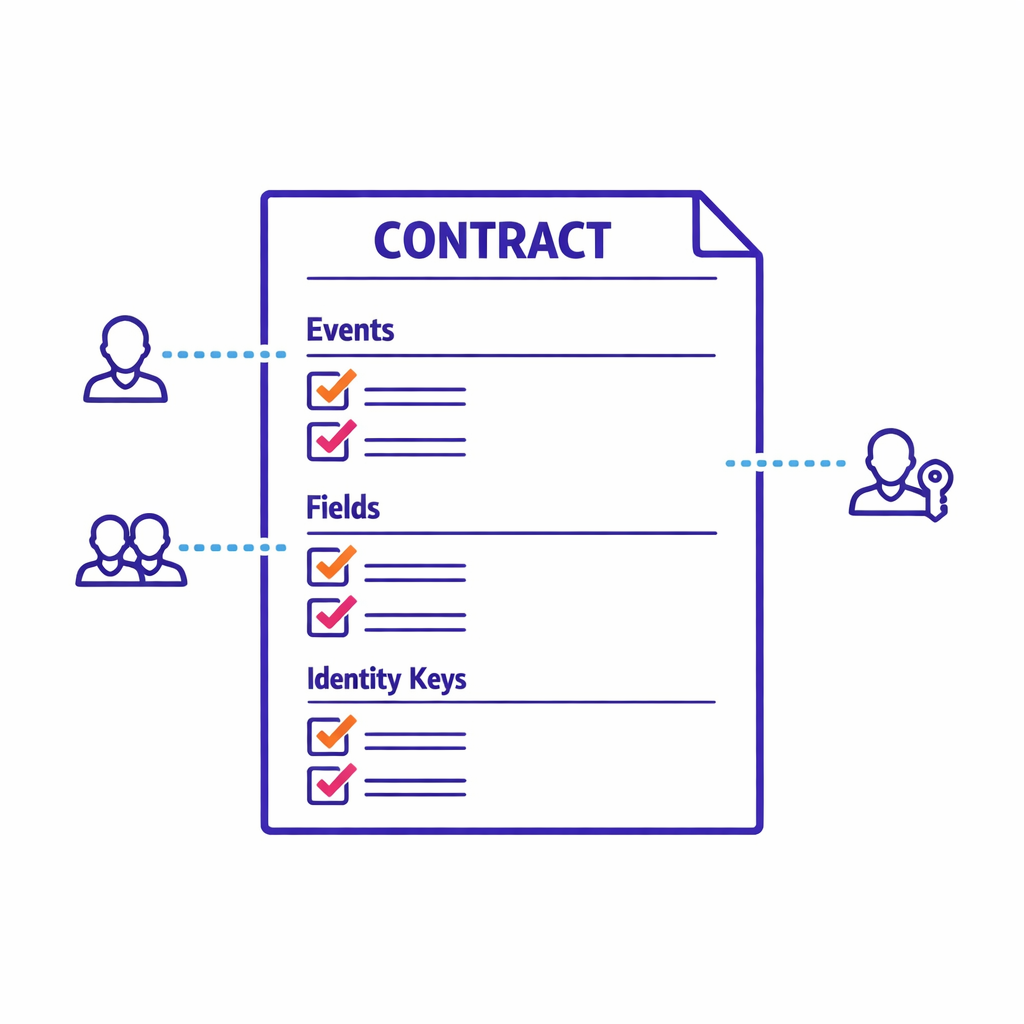
Define a measurement contract partners can implement

Write a partner measurement contract that specifies three things clearly: (1) which events are required, (2) which fields are required for each event, and (3) which identity keys are permitted for alignment. This is where you standardize “engagement” so it means the same thing across HCP and patient programs.
- Event taxonomy: impressions, clicks, site visits, content views, form starts, form completes, enrollments, confirmations.
- Field standards: timestamps, channel, campaign metadata, placement, creative, program ID, consent flags where applicable.
- Identity standards: allowed tokens, NPI where available, and clear rules for when identity is optional vs required.
When partners cannot provide identity keys (common with certain media environments), the contract should still require consistent campaign metadata so you can attribute program-level lift even without person-level joins.
Operational QA: make match quality measurable, not anecdotal
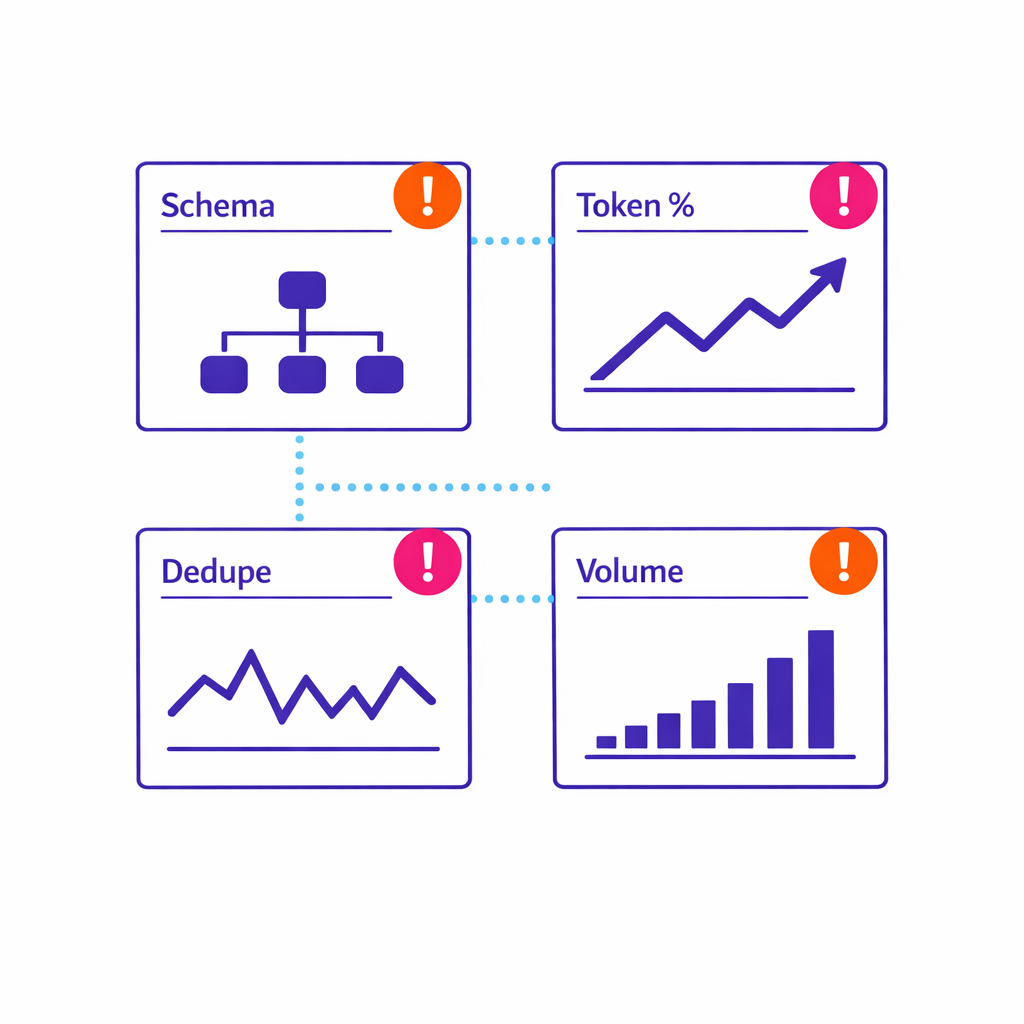
Build QA into the measurement layer so you can evaluate partner feeds continuously. You do not need to overcomplicate it; you need a few standard checks that run every time data lands.
- Schema validation: Are required fields present and properly formatted?
- Token coverage: What percentage of events include a usable token for matching?
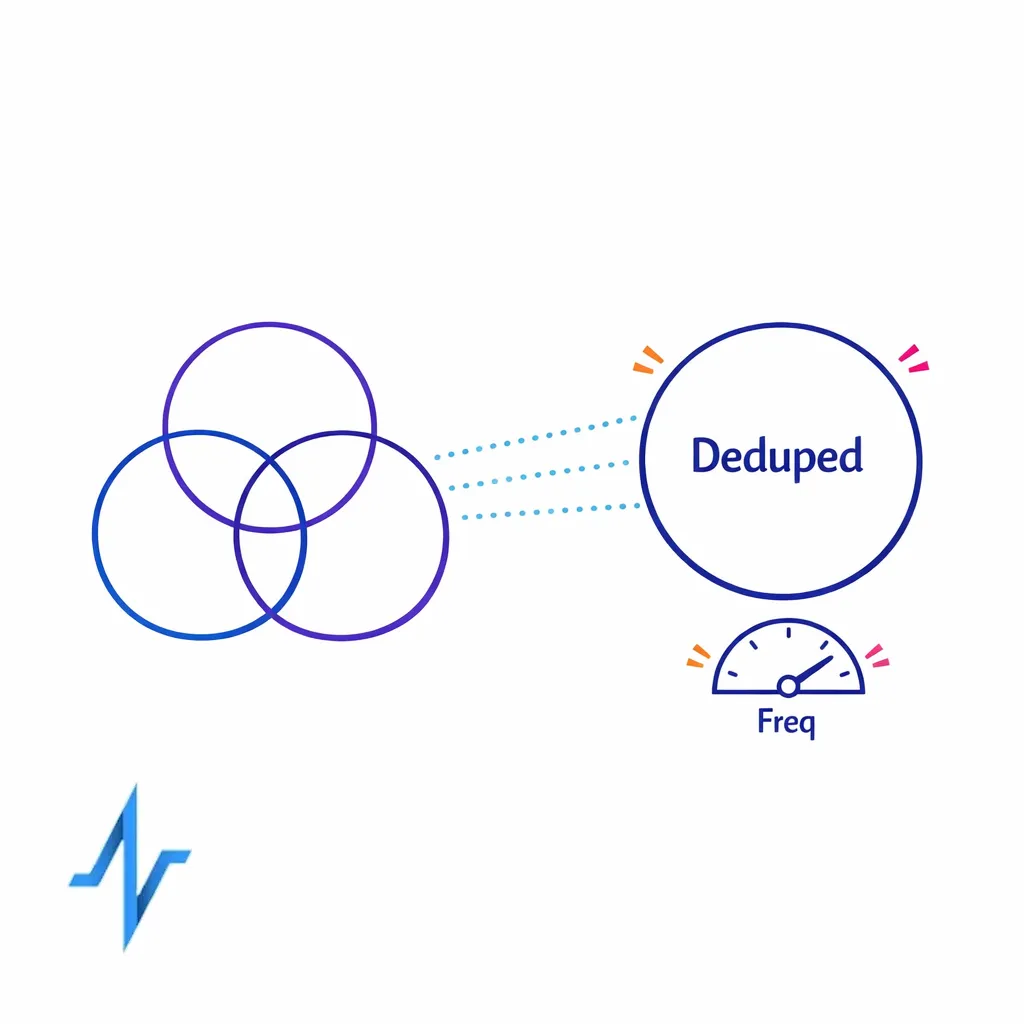
- Deduplication: Are you seeing abnormal repeats or partner-side retries?
- Volume monitoring: Are week-over-week changes explainable by campaign changes?
This is how you turn “data partner alignment” into a managed process rather than a scramble before QBRs.
Measurement outputs: what a de-identified layer should deliver
Once events and identities are aligned, the measurement layer should produce outputs that are useful to brand teams, omnichannel leads, and ops owners. The goal is not a perfect single-touch attribution model. The goal is decision-grade signals, consistently produced, with known limits.
Core omnichannel views for HCP engagement
For HCP programs, your de-identified measurement pharma dataset should support:
- Reach and frequency by segment: How many HCPs did we reach, how often, and in which channels?
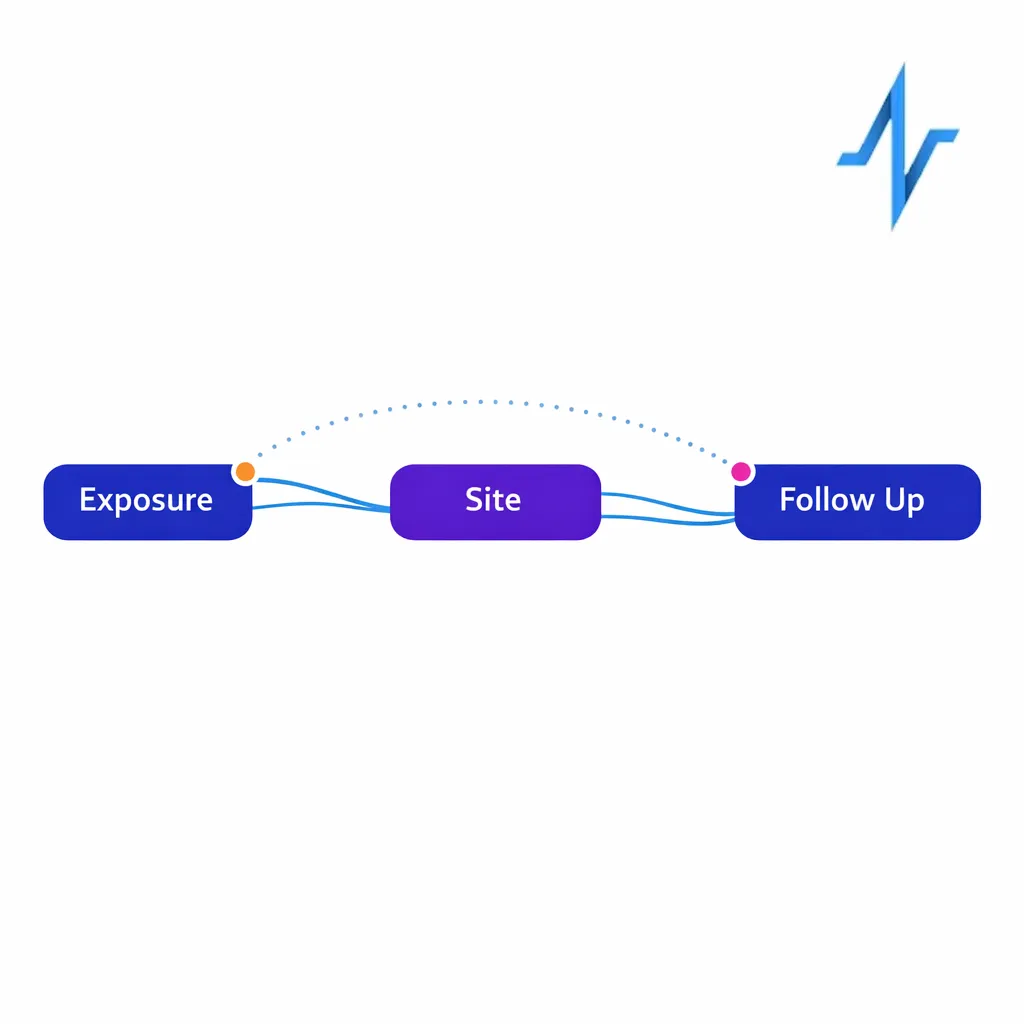
- Journey reporting: Common sequences (e.g., display exposure to site visit to rep follow-up) at an aggregated level.
- Conversion proxies: Form completes, content depth, opt-ins, rep requests, speaker program RSVPs.
Identity resolution pharma teams can operate is the difference between “we think we reached cardiology” and “we can show deduped reach for cardiologists across channels with consistent segment definitions.”
Patient program measurement: acquisition, engagement, and persistence
For patient education and support, measurement needs to connect the top-of-funnel (channel and message) to program engagement without exposing identities. A good measurement layer enables:
- Channel contribution to enrollments: Which sources drive eligible sign-ups and confirmed enrollments.
- Engagement cohorts: How education journeys differ by source, message, or enrollment path.
- Drop-off diagnostics: Where users abandon, by device and step, to inform UX fixes.
Where appropriate, teams can also align program measurement to broader healthcare quality signals that matter to provider organizations. CMS programs like the Hospital Readmissions Reduction Program illustrate how readmissions are treated as a measurable outcome in parts of the system, and CMS’s HCAHPS patient experience survey program shows how patient experience is measured and reported in hospital settings. Your brand program will not “own” these metrics, but building disciplined measurement makes it easier to speak credibly about how education and support efforts are designed to improve patient outcomes meaningfully.
Cross-channel attribution pharma teams can trust (by using the right level of certainty)
In privacy-constrained environments, the most reliable attribution is often a combination of: (1) deterministic matching where permitted, (2) strong campaign metadata everywhere, and (3) experimentation where it is feasible. Use person-level joins for what they are good at (deduplication, frequency, pathing), and use incrementality methods when you need causal answers.
A practical rule: if a decision is high-stakes (reallocating major spend, changing channel mix, changing enrollment workflows), prefer lift-style evidence over last-touch reports, especially when partner data visibility differs by channel.
What changed and what is new: why “cookieless” is now a measurement architecture issue

Pharma measurement has been moving toward first-party and consent-based measurement for years, but platform changes have accelerated the need for a de-identified measurement layer. On mobile, for example, user permission governs certain types of tracking under Apple’s App Tracking Transparency framework, which pushes teams to rely more on first-party identifiers, server-side event collection, and partner alignment rather than third-party cookies alone.
The practical implication for omnichannel leads is straightforward: you need an identity strategy that survives channel policy changes. That usually means investing in tokenization, identity resolution pharma workflows, and partner data alignment as durable capabilities, not one-time workstreams.
Common mistakes and misconceptions (and how to avoid them)

Misconception 1: “Hashing equals de-identification”
Hashed identifiers pharma teams use for joining are helpful, but they are not a substitute for a de-identification strategy. De-identification depends on the standard you are operating under and the total context of the dataset, as reflected in HIPAA’s de-identification provisions in 45 CFR 164.514. Treat hashing and tokenization as privacy controls inside a larger governance model, not as a blanket guarantee.
Misconception 2: “If we have NPI, identity resolution is done”
NPI-based identity is powerful, but it does not automatically solve affiliation hierarchy, account rollups, territory mapping, or attribute freshness. Use NPI as a stable anchor, per CMS’s NPI standard overview, and design your measurement layer to report at multiple levels without forcing everything into one ID.
Misconception 3: “We can just add up partner dashboards”
Summing partner reports almost always double-counts reach and hides frequency problems. Without a consistent identity resolution and event taxonomy, partner dashboards are describing different universes. Align data upstream so reporting downstream is a reconciliation exercise, not a negotiation.
Misconception 4: “Attribution needs a single source of truth”
In regulated, privacy-constrained contexts, a single all-seeing dataset is rarely feasible or desirable. A better target is “fit-for-purpose truths”: separate views for deduped reach, for journey patterns, for conversion rates, and for incrementality. You will make better decisions with three solid lenses than one fragile model.
A practical build plan for a de-identified measurement layer
The fastest path is to build a minimum viable measurement layer that handles identity and partner alignment for a small set of high-value programs, then expand. The steps below are designed to be owned jointly by omnichannel, ops, analytics, and compliance stakeholders.
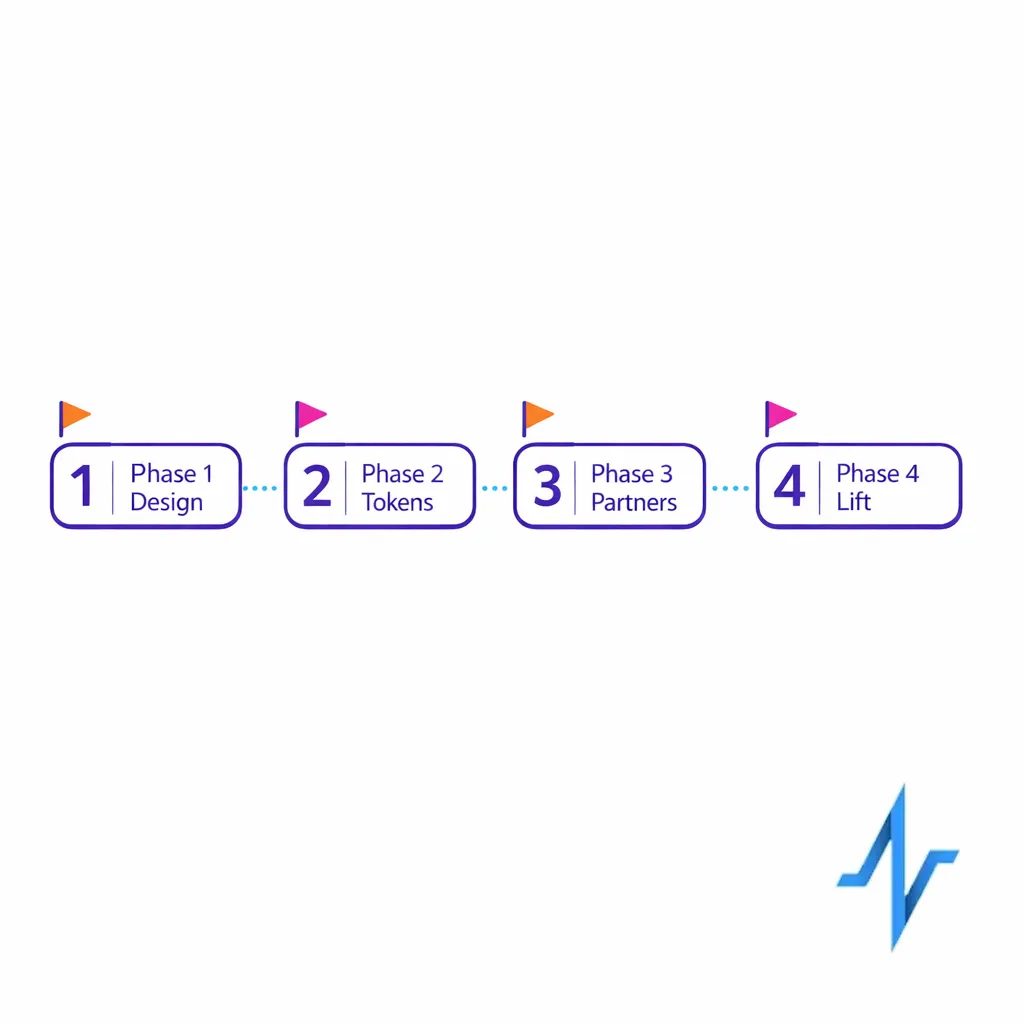
Phase 1 (weeks 0–4): lock the measurement design
- Define the use cases: HCP engagement measurement, patient program measurement, cross-channel attribution pharma reporting, partner performance management.
- Define identity levels: HCP, account, patient enrollment, household (only if justified), program.
- Define event taxonomy: what counts as an exposure, an engagement, a conversion, and a qualified enrollment.
- Define governance: who can access raw inputs, who can access tokens, and what is only available in aggregated form.
Phase 2 (weeks 4–8): implement tokenization and baseline identity resolution

Implement a tokenization service: deterministic where partner alignment is required, and purpose-bound to reduce misuse.
- Implement a tokenization service: deterministic where partner alignment is required, and purpose-bound to reduce misuse.
- Stand up HCP identity matching: map partner HCP feeds to your measurement identity backbone, using NPI when available.
- Build QA and monitoring: schema checks, token coverage, dedupe logic, and volume alerts.
Phase 3 (weeks 8–12): partner onboarding and standardized reporting

Onboard priority partners: agency ad ops feeds, program sites, email vendor, hub enrollments, CRM touchpoints.
- Onboard priority partners: agency ad ops feeds, program sites, email vendor, hub enrollments, CRM touchpoints.
- Publish a reporting layer: deduped reach, frequency, journeys, and conversion views aligned to your taxonomy.
- Document assumptions: where identity joins are deterministic vs inferred vs not available.
Phase 4 (ongoing): add incrementality and outcome-aligned measures

Introduce lift testing where feasible: geo tests, holdouts, or channel-level experimentation depending on constraints.
- Introduce lift testing where feasible: geo tests, holdouts, or channel-level experimentation depending on constraints.
- Align program KPIs to outcomes logic: define intermediate outcomes (education completion, refill reminders engagement) that plausibly connect to improved patient outcomes and operational goals.
- Continuously improve match quality: partner feedback loops driven by QA metrics, not anecdote.
What to do next: a checklist for brand, ops, and agency teams

Use this as a working list for your next measurement architecture review. You should be able to answer “yes” to most of these before you trust omnichannel reporting at scale.
- We have a written event taxonomy used across HCP engagement and patient education/sign-ups.
- We can dedupe reach across at least two major channels using a governed tokenization approach.
- We can explain our identity resolution logic (HCP identity matching rules, patient enrollment identity rules) to internal stakeholders.
- We have partner data alignment standards (fields, formats, cadence, QA checks) that agencies and vendors follow.
- We separate measurement from activation through access controls and purpose-bound identifiers.
- We can produce decision-grade outputs for reach/frequency, journeys, conversions, and lift where feasible.
- We can onboard a new partner in weeks, not quarters because the measurement layer is standardized.
Request a Demo to see a de-identified measurement layer in action

If you are building or modernizing de-identified measurement pharma capabilities, it helps to see how tokenization, identity resolution, and partner data alignment operate as a single system. Request a Demo to walk through how Pulse Health supports measurement architecture for HCP engagement and patient program measurement, including standardized partner feeds, deduped omnichannel reporting, and privacy-by-design analytics.
If you want to pressure-test your current approach first, you can also Book a Consultation to review your identity strategy, reporting gaps, and what a realistic implementation plan looks like across brand, ops, and agency teams. To get oriented before that discussion, you can get the platform overview at Pulse Health and come prepared with your highest-priority channels and partners.






