US pharma teams are being asked to prove impact faster, across more channels, with tighter governance than ever. At the same time, campaign performance tracking often lives in separate systems (media platforms, CRM, webinars, brand sites), which makes “real-time” feel out of reach.
This article is for pharma brand teams, omnichannel leads, commercial ops, marketing ops, CRM owners, and agency partners who need practical pharma marketing analytics that can stand up to day-to-day decisioning and compliance review. You will learn a KPI framework you can standardize, how omnichannel attribution works in regulated contexts, and what a GA4 integration can add to your measurement stack.
Why real-time dashboards are hard (and still worth it) in pharma

“Real-time dashboards” in pharma are less about seeing a number update every second and more about reducing decision lag. When your teams can spot a content drop-off, a form abandonment spike, or a territory-level engagement gap early, you can adjust creative, routing, and orchestration before budget and momentum are gone.
The challenge is that pharma measurement has more constraints than many consumer categories. Governance requirements and partner ecosystems can create fragmented instrumentation, slower data flows, and inconsistent definitions of what counts as an “engagement” or “conversion.”
It also helps to separate two realities that often get mixed together: non-personal promotion measurement (aggregated, channel-level insights) versus person-level signals used for orchestration. Your analytics approach should explicitly define which use cases are allowed to be identity-based and which must remain aggregated, then architect data accordingly.
A KPI framework that works for omnichannel pharma marketing analytics
The fastest way to improve pharma marketing ROI is to standardize a KPI language that every partner can implement the same way. Instead of “every channel reports its own metrics,” align metrics to a small set of outcomes and instrument them consistently across touchpoints.
1) Exposure and reach (prove you showed up)
Use these to understand whether your mix is actually delivering opportunity to engage. They are especially important when your goal is to improve efficiency (reach the right audience with less waste) rather than only driving downstream actions.
- Reach and frequency by channel and audience segment (as available from each platform).
- Share of voice proxies in paid channels (impressions, auction insights, or placement coverage where applicable).
- Landing page coverage: percent of campaigns with a live, tagged destination and working measurement.
2) Content engagement analytics (prove the experience worked)
Pharma content often does the heavy lifting: communicating disease education, MOA storytelling, safety framing, and next-step guidance. “Engagement” should not be a single number. Define a tiered ladder so you can diagnose where users drop off.
- Quality visits: sessions with meaningful depth (for example, multiple content interactions or time on key modules).
- Module completion: video completes, scroll depth milestones, carousel steps, or interactive tool completion.
- Return engagement: repeat visits to high-value education content within a defined window.
Operationally, the goal is to move from “what got clicks” to “what advanced understanding and intent.” A consistent event taxonomy (the same event names and parameters across experiences) is what makes this comparable across agencies and properties.
3) Intent and next-step actions (prove momentum)
For HCP engagement and patient education/sign-ups, define “next-step” actions that are legitimate indicators of progress without over-claiming clinical outcomes. These should be designed with medical, legal, and regulatory stakeholders so the KPI set is defensible.
- HCP pathways: formulary or access resource views, rep contact requests, sample requests (where applicable), or “learn more” sequences that indicate clinical interest.
- Patient pathways: education hub progression, eligibility checks, program interest submissions, or support resource interactions.
- Lead hygiene: percent of submissions that pass validation, dedupe rate, and time-to-routing for follow-up.
4) Orchestration and operations (prove the system is working)
Many omnichannel programs fail quietly due to operational friction, not creative quality. Marketing ops and commercial ops should measure the health of the system that turns engagement into coordinated action.
- Audience freshness: how quickly segments update and how often audiences drift out of policy-compliant eligibility criteria.
- Time-to-activate: time from signal to downstream action (for example, trigger to email send, trigger to rep task creation, trigger to content recommendation).
- Coverage and leakage: percent of high-intent users who receive the next best action versus falling into “no journey” gaps.
Real-time dashboards: what “real-time” should mean in pharma
In practice, “real-time” is a set of service-level expectations you define, not a single technology choice. Decide which decisions require intraday visibility (for example, paid media pacing and creative fatigue) versus daily visibility (for example, content sequencing and nurture performance) versus weekly visibility (for example, mix optimization across channels).
A real time analytics dashboard becomes genuinely useful when it answers three questions quickly: What changed, why did it change, and what should we do next. That means dashboards should be built around actions (pause a creative, swap a landing page module, adjust an audience rule), not around vanity KPIs.
Dashboards that tend to perform well
- Campaign performance tracking cockpit: spend, delivery, qualified engagement, and next-step actions with clear pacing signals.
- Content engagement analytics view: module-level drop-off, completion, and pathway analysis to spot where education stalls.
- Journey and orchestration view: trigger volumes, send volumes, suppression reasons, and handoff timing (especially for CRM owners).
- Geo/territory diagnostic: aggregated engagement patterns mapped to operational levers (coverage, routing, field coordination).
Omnichannel attribution in pharma: what it is, what it is not
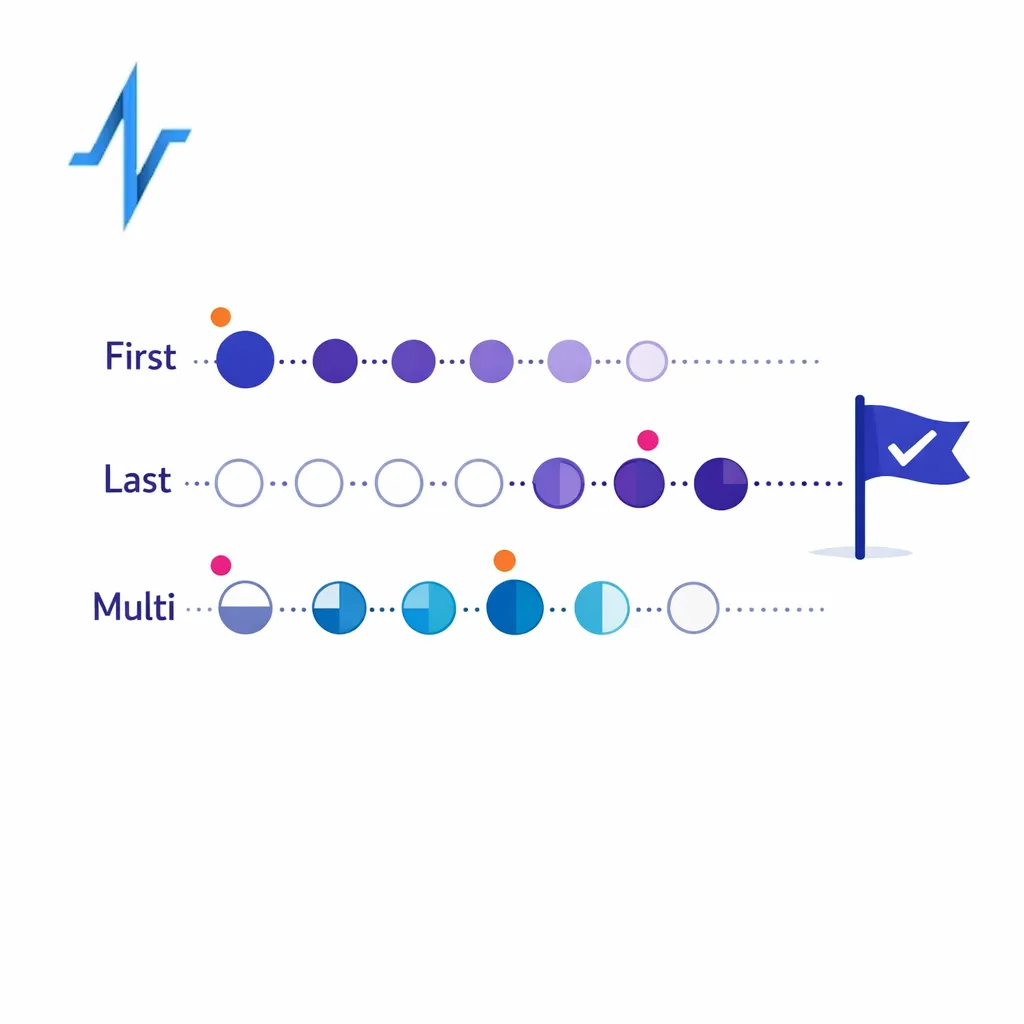
Omnichannel attribution is the practice of assigning credit for an outcome across multiple touchpoints. In pharma, the right approach depends on what you are measuring: non-personal promotion measurement, person-level orchestration, or business outcomes that are influenced by many factors beyond marketing.
Attribution is also not a single model. You will typically use a portfolio of approaches depending on the decision you are making. The key is to be explicit about limitations, bias, and what the model is allowed to claim.
Common attribution approaches (and when they help)
- Rules-based attribution (first touch, last touch, linear): good for lightweight directional learning and consistent reporting definitions.
- Multi-touch attribution (MTA): useful when you have reliable, policy-compliant identity signals and enough volume to learn patterns.
- Incrementality testing: best when the question is “did marketing cause lift,” not “which touch gets credit.”
- Marketing mix modeling (MMM): helpful for longer horizons and channels where user-level tracking is limited.
Two governance points matter in pharma: what user-level data you are permitted to use for modeling, and how results will be reviewed and explained. If your program includes prescription drug promotion, teams often align measurement governance with the oversight expectations associated with the FDA Office of Prescription Drug Promotion (OPDP), even when analytics itself is not promotional content.
Identity + measurement: keep the “how” separate from the “who”
Attribution improves when you can connect touchpoints across devices and channels, but the way you do that must match your privacy posture and program type. A practical pattern is to keep a clean separation between (1) aggregated measurement that supports non-personal promotion decisions and (2) permitted identity workflows used for orchestration in approved systems.
When analytics intersects with regulated data types, your compliance stakeholders may look to frameworks like the HIPAA Privacy Rule to clarify responsibilities for covered entities and business associates. Even when HIPAA is not directly applicable, the discipline of documenting allowed uses, data flows, and access controls is still valuable.
What GA4 adds to pharma marketing analytics (and what it does not)
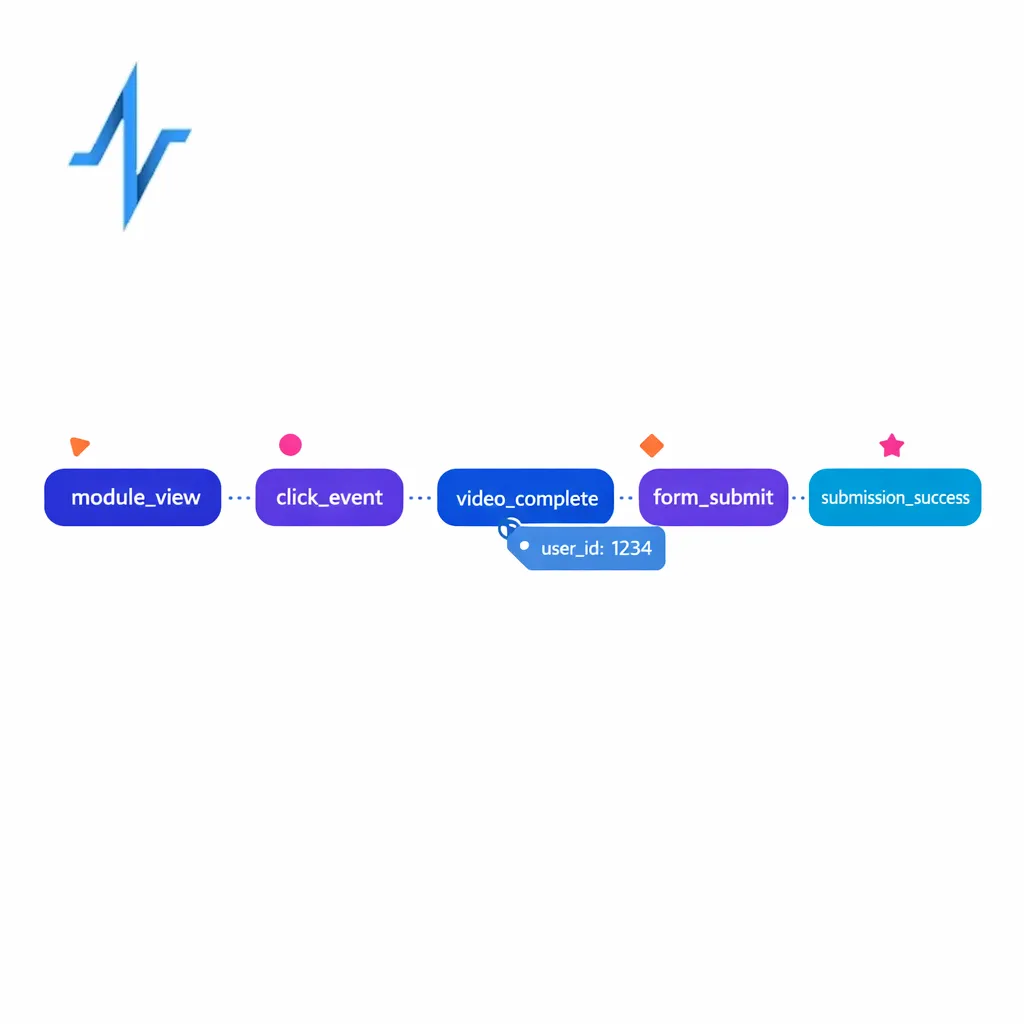
GA4 is not a full omnichannel measurement system by itself, but it can be a strong foundation for digital experience analytics when implemented carefully. The biggest value comes when GA4 is treated as an event stream you can standardize, combine with other sources, and operationalize in dashboards.
GA4’s event-based model makes KPI standardization easier
Unlike older pageview-first approaches, GA4 is built around events and parameters, which is useful for pharma sites with modular content and multiple “micro-conversions.” You can use GA4’s event measurement model to define consistent engagement signals across brand sites, education hubs, and campaign landing pages (for example, module_view, safety_expand, video_complete, eligibility_start, submission_success).
This helps marketing ops teams reduce agency-to-agency variation. Instead of arguing about whether “engagement” means bounce rate, time, or clicks, you implement explicit events and measure the same thing everywhere.
GA4 supports attribution reporting that can complement your omnichannel models
GA4 includes attribution reporting and model options that can help you understand how digital touchpoints contribute to conversions. For teams evaluating channel roles and pathing, GA4’s attribution models and attribution reporting can provide directional insights that you can compare against other methods (like tests or mix models) rather than treating it as the single source of truth.
In pharma, the most sustainable posture is to treat GA4 attribution as one lens. Use it to improve digital experience decisions and paid-to-owned flow efficiency, while reserving broader “business impact” claims for methods designed to answer causal questions.
GA4 can make “near real-time” experience monitoring more accessible
For rapid diagnostics during launches or creative rotations, GA4 provides a Realtime report that helps teams see current activity, top pages, and event activity as instrumentation is rolling out. This is especially helpful for catching broken tags, misfiring conversions, or sudden traffic quality issues early.
For pharma teams, a useful operating model is to use real-time views for validation and incident response, then use daily and weekly views for optimization decisions that require stable denominators.
GA4’s export options help you build a unified measurement layer
Many teams hit a ceiling when analytics data is trapped inside a UI. GA4 can export raw event data to BigQuery, which supports building a more flexible measurement layer for dashboards, modeling, and data quality controls. GA4’s BigQuery Export is often the bridge between website/app engagement signals and enterprise reporting environments.
For commercial ops, this matters because your most actionable reporting typically combines multiple sources: media delivery, site engagement, CRM journey performance, and (where permitted) downstream operational outcomes. A warehouse-centered approach also enables consistent metric definitions across brands and agencies.
GA4 integration: a practical implementation map for pharma teams
A successful GA4 integration is less about turning on tracking and more about designing a measurement system your organization can govern. Start with a blueprint: the KPI definitions, the event taxonomy, the conversion logic, and the handoffs into dashboards and orchestration tools.
Step 1: Define your measurement charter
Document what decisions the dashboards will drive. Tie each decision to an observable signal, an owner, and a cadence. This keeps the project grounded in outcomes and reduces the temptation to track everything “just in case.”
- Decisions: pacing, creative rotation, content edits, journey tuning, audience refinement.
- Cadence: intraday validation, daily optimization, weekly planning, monthly performance reviews.
- Owners: brand, media, CRM, analytics, compliance stakeholders.
Step 2: Build an event taxonomy that matches pharma journeys
Event design is where content engagement analytics becomes real. Standardize naming, required parameters, and how you identify content modules. Keep it modular so new experiences can reuse the same instrumentation.
- Core events: content_view, module_start, module_complete, outbound_click, search_use.
- Safety-aware events: safety_expand, safety_link_click, important_info_view (as applicable to your experience design).
- Form funnel events: form_start, step_progress, validation_error, submission_success.
Make sure the taxonomy distinguishes between “user did something” and “system did something.” That separation is critical when you later connect site events to CRM actions and want to avoid double-counting.
Step 3: Align conversion definitions to the job-to-be-done
In pharma, the “conversion” is often a proxy action, not a purchase. Align conversions to the business purpose of the experience and validate with stakeholders: education completion, support interest, HCP follow-up request, or other approved next steps.
Design conversions to be resilient to channel mix changes. If you change your paid strategy, the conversion definition should not break. The easiest way to accomplish this is to base conversions on the standardized events, not on brittle page URLs.
Step 4: Put data quality checks into the launch process
Real-time monitoring is most valuable as a quality gate. Before traffic scales, validate that events fire, parameters populate, conversions count once, and dashboards reconcile against platform totals within expected tolerance.
- Tagging validation: confirm key events appear and include the required fields.
- Funnel validation: ensure each funnel step can be observed and is ordered correctly.
- Reconciliation: compare sessions, clicks, and conversions across systems with documented assumptions.
Common mistakes and misconceptions (and how to avoid them)
Teams sometimes chase second-by-second reporting when the real need is operational responsiveness. Decide what must be fast versus what must be accurate and stable, then build dashboards to match those decision cycles.
Mistake 1: Treating “real-time” as the goal
Teams sometimes chase second-by-second reporting when the real need is operational responsiveness. Decide what must be fast versus what must be accurate and stable, then build dashboards to match those decision cycles.
Mistake 2: Measuring channels instead of journeys
Channel dashboards often encourage channel-optimized behavior. Journey dashboards encourage outcome-optimized behavior. If your KPI set does not carry from paid to owned to CRM, you will struggle to explain performance and you will waste effort reconciling reports.
Mistake 3: Assuming attribution is the same as incrementality
Attribution assigns credit based on observed paths. Incrementality asks whether an outcome would have happened without a tactic. Treat attribution as directional learning and use testing or other causal approaches when you need “did this cause lift” answers.
Mistake 4: Over-instrumenting and under-governing
More events do not automatically create more insight. If every agency invents its own parameters, your GA4 integration becomes a noisy data lake instead of a measurement system. A smaller, governed event taxonomy usually produces faster and more trustworthy reporting.
Mistake 5: Believing GA4 is the omnichannel source of truth
GA4 is excellent for digital experience analytics, but omnichannel attribution typically requires combining multiple systems (media, CRM, call center, webinar platforms, field activity) with clear identity and governance boundaries. Use GA4 as a core input, then unify it with the rest of your stack in a controlled reporting layer.
What to do next: a practical checklist for brand teams and ops owners
If you want real-time dashboards that drive decisions, treat the next 30–60 days as a measurement enablement sprint. Focus on definitions, instrumentation, and a small set of dashboards that map to actions.
- Standardize KPIs: agree on 10–20 cross-channel KPIs and document precise calculation rules.
- Define your event taxonomy: pick a small set of required events and parameters that every site and landing page must implement.
- Design “conversion” proxies: align conversions to approved next-step actions for HCP engagement and patient education/sign-ups.
- Implement GA4 with governance: enforce naming conventions, QA checklists, and version control for tagging changes.
- Choose an attribution posture: decide which questions you will answer with rules-based attribution versus testing or mix modeling.
- Build two dashboard tiers: (1) intraday validation and incident response, (2) daily/weekly optimization and planning.
- Operationalize follow-through: assign owners and response playbooks (what happens when a KPI goes red).
Talk to Pulse Health
If your team needs pharma marketing analytics that are fast enough for day-to-day optimization and structured enough for governance, Pulse Health can help you unify signals across channels, build real-time dashboards that teams actually use, and connect measurement to orchestration without creating reporting chaos.
Start with one high-impact use case (like campaign performance tracking for a launch, or content engagement analytics for a key education hub), then expand. You can request a demo with Pulse Health, book a consultation to map your KPI and attribution approach, or explore integrations to see how GA4 integration can fit into your existing stack.



