Pharma teams are being asked to deliver more personalized, measurable engagement across HCP and patient journeys while tightening governance around data, content, and vendor risk. A “pharma engagement cloud RFP” is where those expectations become real requirements, and where unclear scope can quickly turn into integration rework, compliance friction, and underused capabilities.
This article is for US pharma brand teams, omnichannel leads, commercial ops, marketing ops, CRM owners, and agency partners who need a practical, compliance-aware vendor evaluation. You will get a requirements checklist, ready-to-use RFP questions, and a scoring framework that brand, ops, and IT can align on in 2026.
How to use this checklist (before you write the RFP)
A strong RFP starts with a shared definition of the problem and the operating model. The goal is not to list every feature you can imagine, but to define what “good” looks like in outcomes, workflows, governance, and measurable adoption.
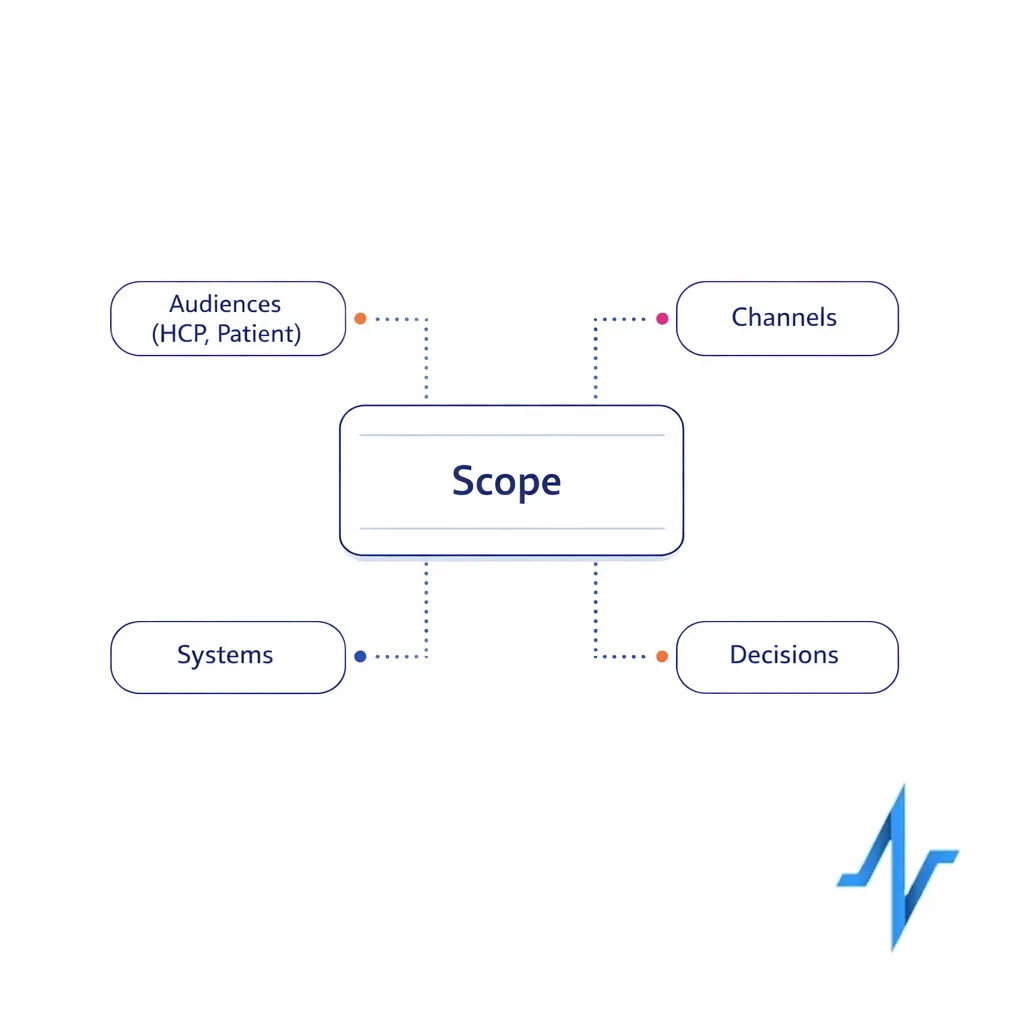
Define the engagement cloud scope in one paragraph
Write a short scope statement that covers: (1) who you engage (HCPs, patients, caregivers), (2) which channels you orchestrate, (3) what systems you must integrate with, and (4) what decisions the platform should enable (next best action, segmentation, measurement, or content operations). If you cannot summarize scope clearly, scoring will become subjective and implementation will drag.
Align on a small set of measurable outcomes
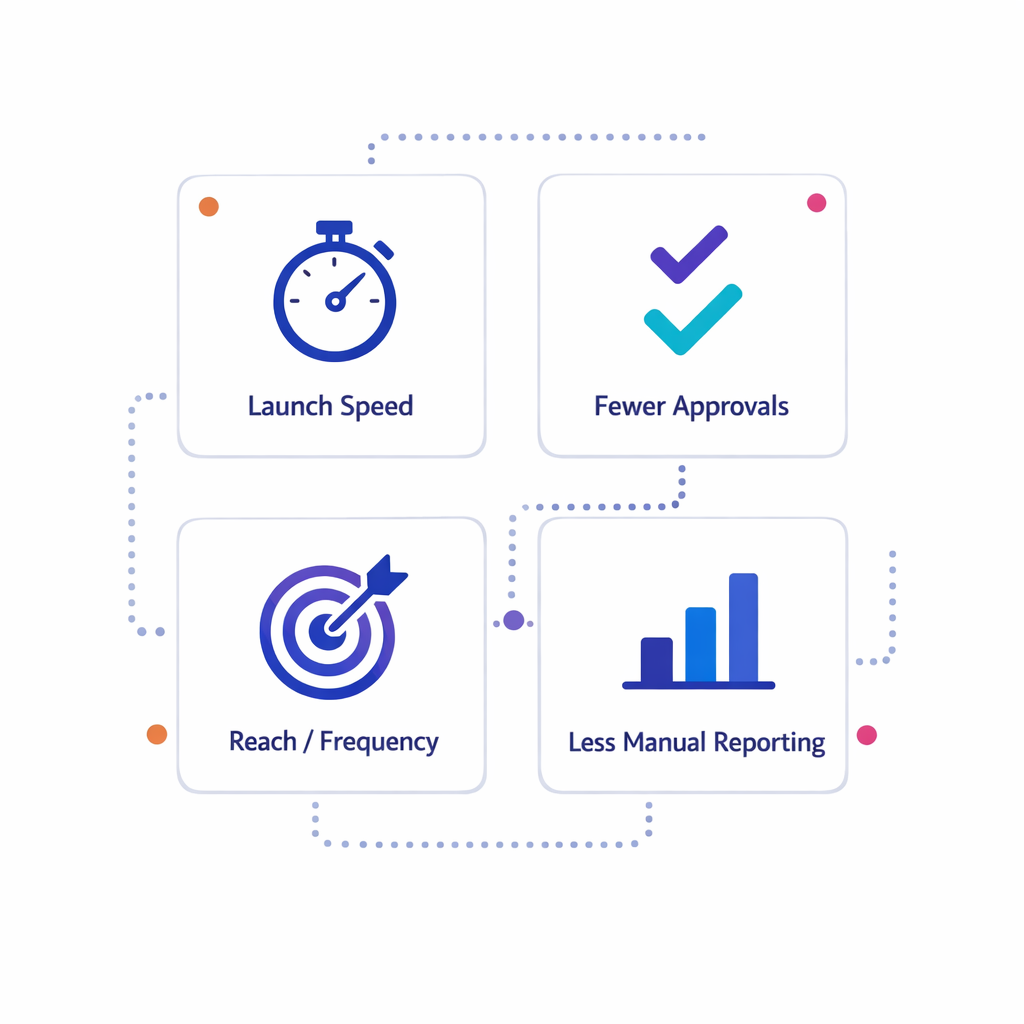
Pick 4–6 outcomes you will use to evaluate the platform after go-live. Keep them operational and measurable, such as faster campaign launches, fewer handoffs for approvals, improved reach frequency to target HCP segments, improved patient program enrollment completion, and reduced manual reporting effort.
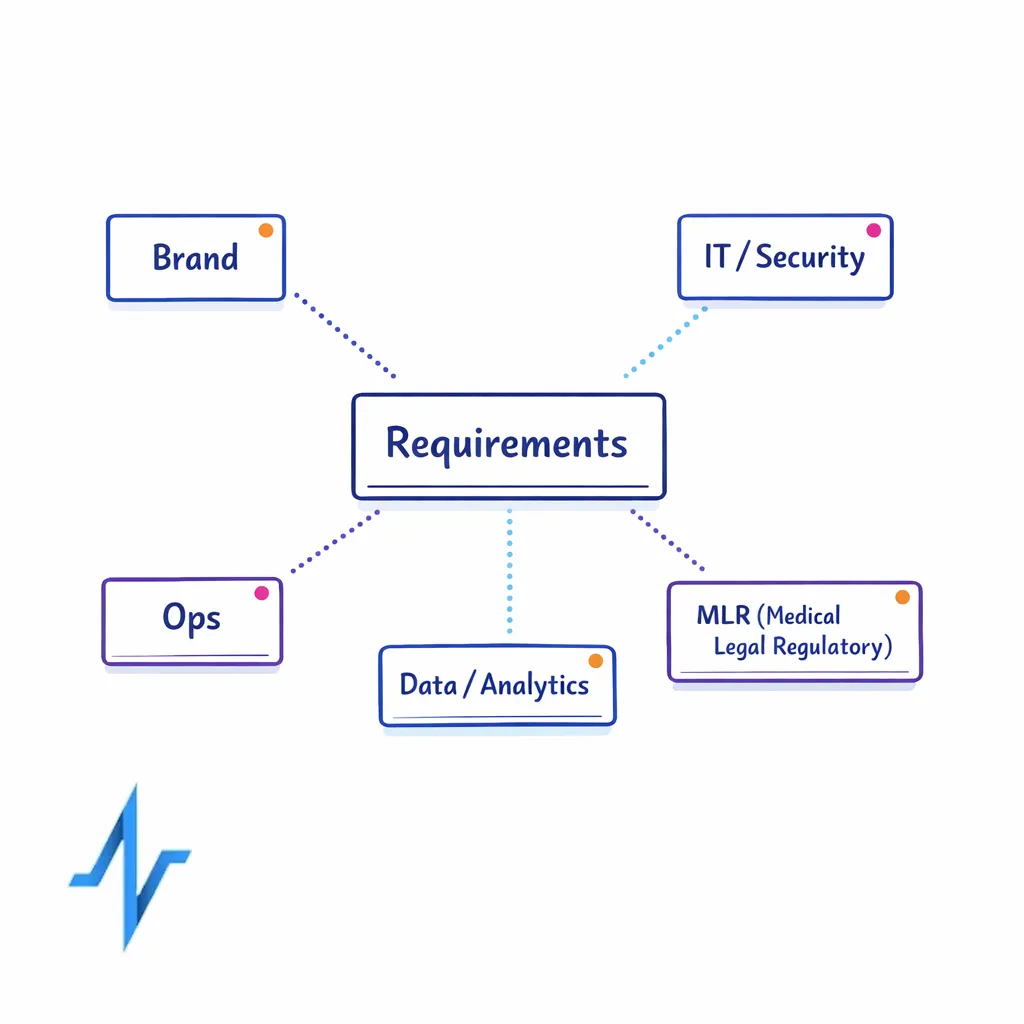
Get the right stakeholders into the requirements workshop
- Brand and omnichannel: journey strategy, channel priorities, message governance, field coordination.
- Commercial ops / marketing ops: CRM process, targeting, campaign ops, tagging, SLAs, training.
- Data / analytics: identity, measurement, attribution approach, data contracts, quality rules.
- IT / security: architecture, integrations, security requirements, vendor due diligence, access controls.
- Medical / legal / regulatory (MLR): content lifecycle and promotional review expectations.
What changed in 2026 vendor evaluations (and why it matters)
RFPs in 2026 are less about “does it have omnichannel features” and more about whether the platform can be operated safely at scale across privacy, security, and auditability expectations. That shift affects how you write requirements, what evidence you request, and how you score risk.
Privacy expectations are broader than HIPAA-only thinking
If your engagement cloud supports patient education, enrollment, or follow-up outside a covered entity context, you may still face breach and notice expectations under the FTC Health Breach Notification Rule. If your programs operate within HIPAA-covered contexts, align requirements with the HIPAA Privacy Rule early, especially around permitted uses, disclosures, and minimum necessary practices.
Security questionnaires increasingly expect recognized frameworks and evidence

Many procurement teams now require proof that security controls are managed under an auditable program aligned to standards like ISO/IEC 27001:2022, mapped to a risk framework such as the NIST Cybersecurity Framework, and supported by third-party attestation such as SOC reports for service organizations. This has practical RFP implications: you should request evidence, not just “yes” answers.
Promotional governance is still a core design constraint
Engagement clouds that touch HCP promotion must support workflows that enable compliant operations across content creation, review, and distribution. When in doubt, your RFP should reflect that prescription drug promotion is overseen by the FDA Office of Prescription Drug Promotion (OPDP) and that auditability and traceability are not optional in real-world teams.
RFP structure: sections, artifacts, and response format
To make vendor responses comparable, specify how you want requirements answered. The fastest path to clarity is to require vendors to provide “how it works” descriptions, implementation assumptions, and evidence links for security and compliance claims.
Recommended RFP sections
- Business goals and use cases: what you are trying to improve, and how success will be measured.
- Functional requirements: orchestration, journeys, content operations, audience management, channel execution.
- Data, identity, and measurement: first-party data strategy, consent, identity resolution, reporting, governance.
- Integration and architecture: CRM, data warehouse, MAP, CMS, consent tools, SSO, event collection.
- Security and vendor due diligence: certifications, controls, IR, DR, pen testing, access and logging.
- Implementation and onboarding: roles, timeline, migration, training, change management.
- Support and SLAs: uptime targets, support model, escalation, release management.
- Commercials: pricing model, overages, services, contract terms, exit plan.
Standardize the answer format
- Require a one-paragraph answer for “what it does” and a one-paragraph answer for “how it is implemented/operated.”
- Require vendors to tag each requirement as Available now, Configurable, Requires services, or Roadmap with dates.
- Require evidence for risk claims (for example, attach SOC report under NDA, describe ISO scope, show sample audit logs).
Functional requirements (brand and omnichannel): what “good” looks like
Brand and omnichannel teams should focus on three areas: orchestration that reflects real operating constraints, personalization that is measurable and governable, and content workflows that fit your MLR reality. Avoid feature shopping and prioritize the workflows you will run weekly.
1) Journey orchestration and campaign operations
- Journey design: ability to define sequences, eligibility rules, exclusions, and caps (frequency, channel limits, quiet periods).
- Operational controls: approvals, scheduling, throttling, and audit trails for changes to targeting and content mapping.
- Experimentation: A/B testing and holdouts with clear reporting definitions and guardrails.
RFP questions for orchestration
- Describe how a marketer builds a multi-step journey with suppression rules and frequency caps. What parts are self-serve versus admin-only?
- How do you prevent conflicting messages across concurrent journeys (for example, brand and patient support programs targeting the same person)?
- How are changes versioned and audited (targeting logic, content mapping, scheduling, exclusions)?
2) HCP engagement and field coordination
Your platform should support coordinated engagement across digital and field motions without forcing the field into disconnected tools. For many teams, the critical capability is not “more channels” but a shared understanding of what happened, what is allowed next, and how to measure incremental lift.
RFP questions for HCP engagement
- What is your model for coordinating with CRM activities (rep touchpoints, speaker programs, samples) without duplicating systems of record?
- How do you manage territory, account, and affiliation concepts so that targeting reflects real sales alignment?
- How do you support compliant content delivery and tracking for HCP communications, including opt-outs and channel preferences?
3) Patient education and sign-up flows
If you support patient programs, the core requirements are clear consent capture, high-quality data collection, and operational visibility into funnel drop-off and enrollment completion. Keep requirements explicit about data ownership, permitted uses, and downstream activation rights.
RFP questions for patient flows
- How do you capture consent and preferences, and how are those preferences enforced across channels and downstream systems?
- How do you handle identity resolution when a patient starts on one device/channel and completes on another?
- What reporting is available for enrollment funnel analytics, and how do you export event-level data for analytics?
Data, identity, and measurement requirements (data governance-first)
Identity and measurement are where many engagement clouds fail, not because the platform is weak, but because requirements were vague. Your RFP should force clarity on data contracts, identity rules, and what measurement is feasible given privacy constraints.
1) First-party data strategy requirements
- Data capture design: forms, event tracking, preference centers, and consent capture patterns.
- Data contracts: defined schemas, required fields, validation rules, and data quality checks.
- Governance: documented ownership of data elements (who can create, modify, approve, and deprecate fields).
RFP questions for first-party data
- Provide a sample data dictionary and event taxonomy you recommend for HCP engagement and patient enrollment programs.
- How do you prevent “shadow fields” and inconsistent tagging across brands, agencies, and business units?
- What tools exist for data quality monitoring (completeness, duplicates, invalid values) and what is automated?
2) Identity resolution and preference enforcement
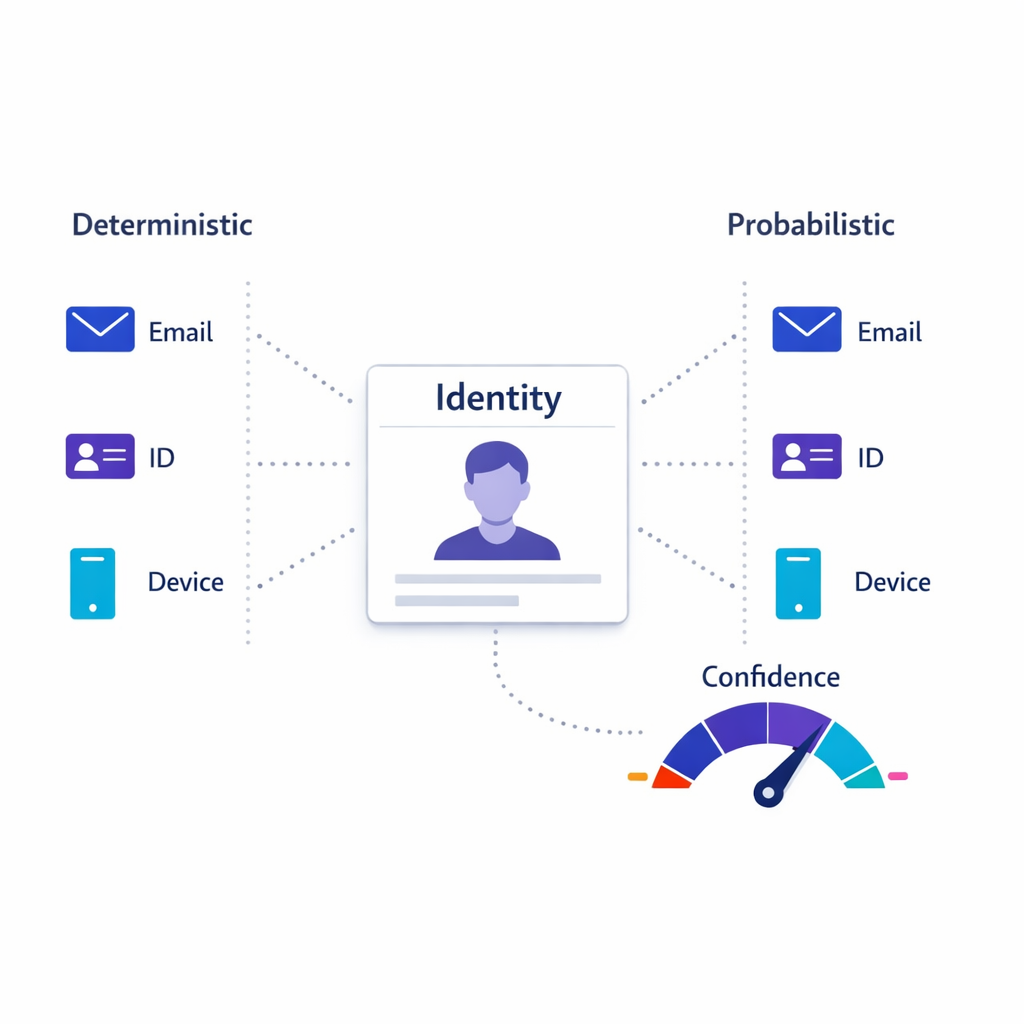
Identity resolution should be described as a set of deterministic and probabilistic rules with transparency, not as a black box. Preference enforcement should be consistent across channels and easy to audit when questions arise.
RFP questions for identity and measurement
- Explain your identity model (identifiers supported, matching approach, survivorship rules, and confidence scoring).
- How are opt-outs and channel preferences stored, time-stamped, and applied in real time?
- How do you provide “explainability” for why an individual was included or excluded from a journey?
3) Analytics and reporting requirements (including Pulse Analytics expectations)
Many teams want self-serve dashboards, but the real requirement is consistent measurement definitions and accessible data for deeper analysis. If you plan to standardize reporting through a layer such as Pulse Analytics, require vendors to show how event data, metadata, and cost inputs become usable metrics across brands.
- Standard metrics: reach, frequency, engagement, conversion, drop-off, time-to-next-step, incremental lift frameworks.
- Data export: event-level export with clear schemas and retention policies.
- Governed reporting: role-based access, approved definitions, and version history of KPIs.
Integration and architecture requirements (ops + IT alignment)
CRM integration requirements and downstream data activation are where platform “fit” becomes real. Your RFP should specify systems in scope, integration patterns you prefer, and operational ownership of monitoring and failures.
1) CRM integration requirements (including Veeva and Salesforce integration considerations)
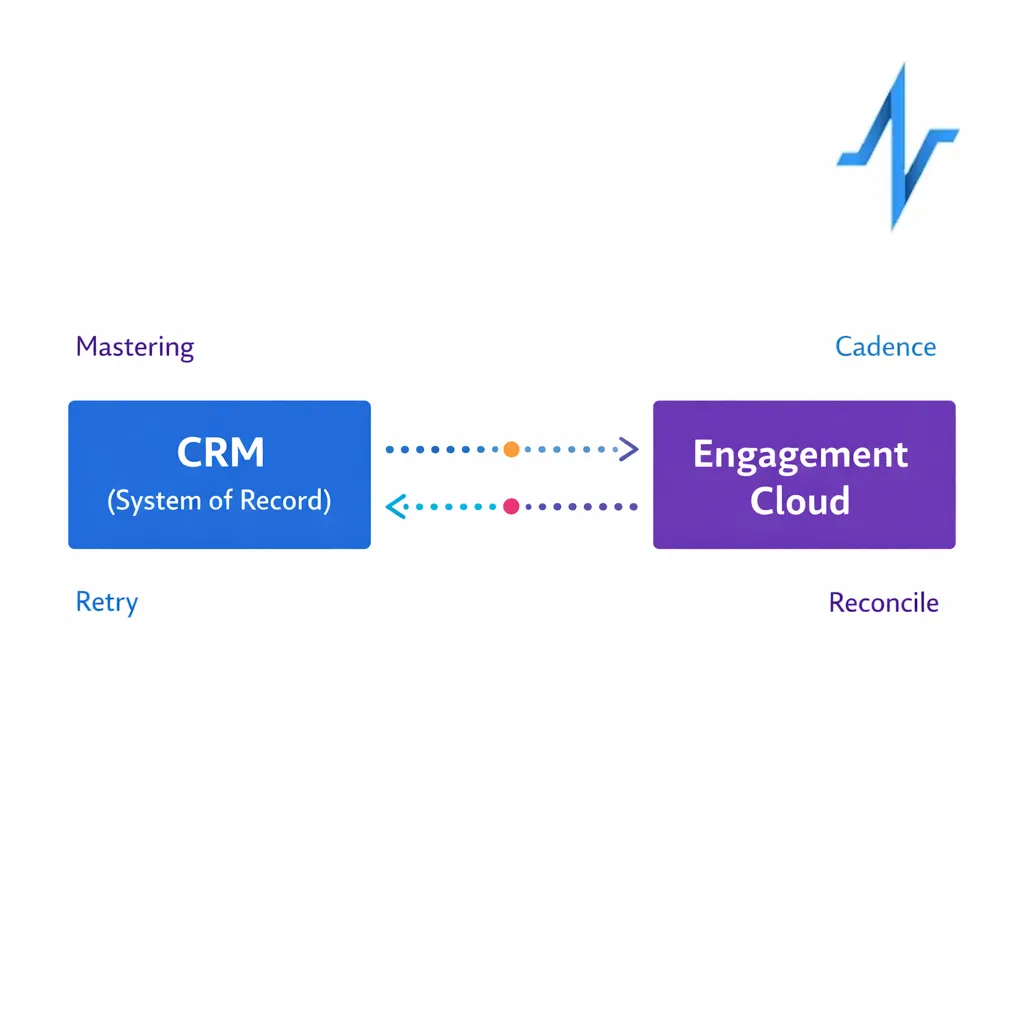
Many pharma organizations run CRM as a system of record for HCP data and field activity, so the engagement cloud should integrate without creating duplicate sources of truth. Your RFP should ask vendors to describe the canonical record approach, sync cadence, conflict resolution, and how they handle territory changes and affiliation updates.
- Data direction: define what is mastered in CRM versus the engagement cloud.
- Sync design: batch, near-real-time, or event-driven, with retry behavior and reconciliation.
- Operational ownership: who monitors failures, where alerts go, and what “fix” playbooks exist.
RFP questions for CRM integrations
- Describe your standard integration pattern for CRM systems (including how you handle deduplication, conflict resolution, and record locking).
- What are your recommended environments (dev/test/prod) and how do you support sandbox refreshes and test data management?
- How do you support agency partners while maintaining least-privilege access to CRM-connected data?
2) Data warehouse, BI, and activation integrations
Assume that not all analytics will live in the engagement cloud UI. Require event-level data availability, clear data dictionaries, and support for feeding your enterprise warehouse and BI tools so reporting can be standardized across brands and partners.
RFP questions for data and BI
- Can you deliver event-level exports with stable schemas and late-arriving event handling?
- How do you ensure metric consistency across the UI and exported datasets?
- How do you support cost ingestion (media, program costs) for ROI reporting and budget governance?
Compliance and governance requirements (make them operational)
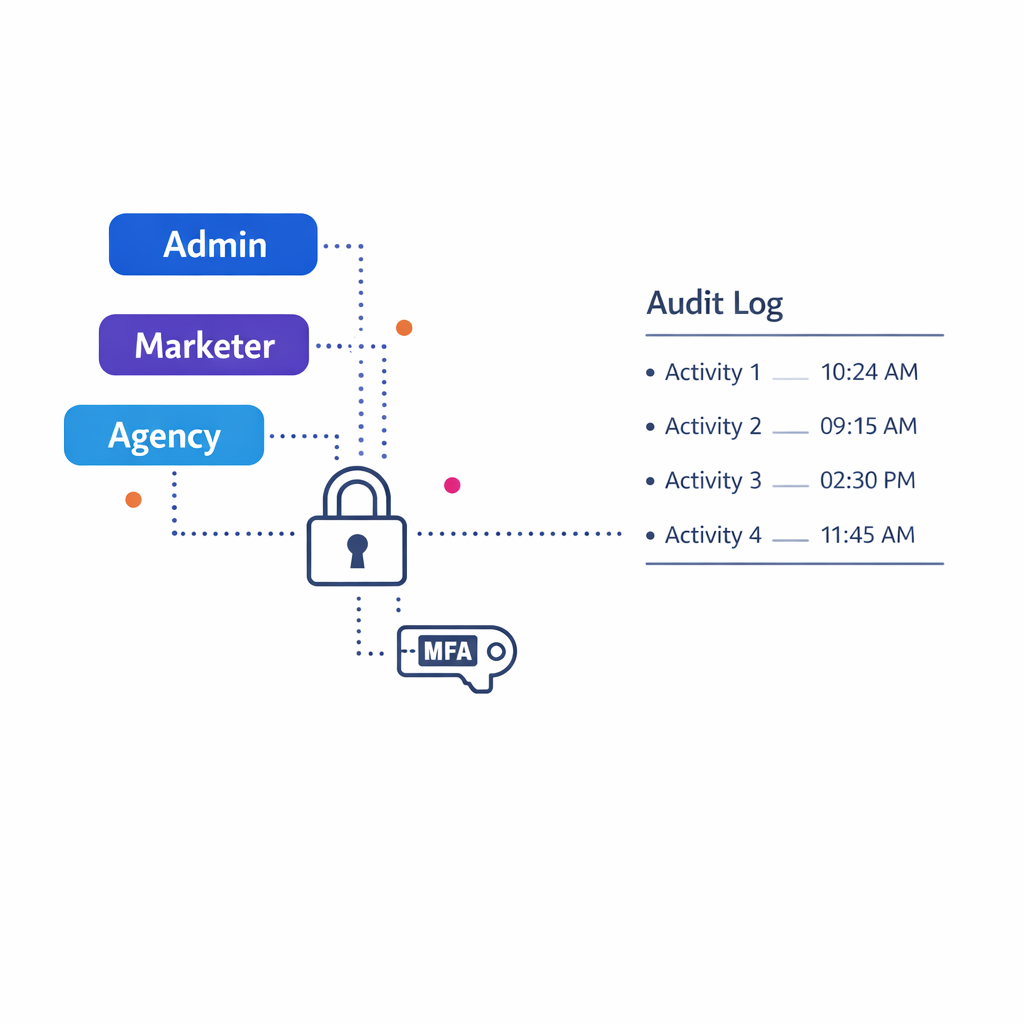
In pharma, “compliance” is not a checkbox. It is a set of operational behaviors: content governance, access controls, audit trails, and clear data use boundaries across vendors and partners.
1) Privacy and health data handling
If a vendor touches protected health information in a covered entity context, align requirements to the HIPAA Privacy Rule and require clarity on when the vendor is acting as a business associate, what data is in scope, and what safeguards are in place. If your patient education or enrollment experiences are outside HIPAA, include expectations aligned to the FTC Health Breach Notification Rule, including incident response and consumer notice workflows where applicable.
RFP questions for privacy
- What data types do you classify as sensitive, and how is sensitive data handled differently (storage, access, logging, masking)?
- How do you support data minimization and “minimum necessary” operational practices?
- Describe your incident response workflow, including customer notification timelines and what evidence you provide post-incident.
2) Promotional review readiness and auditability
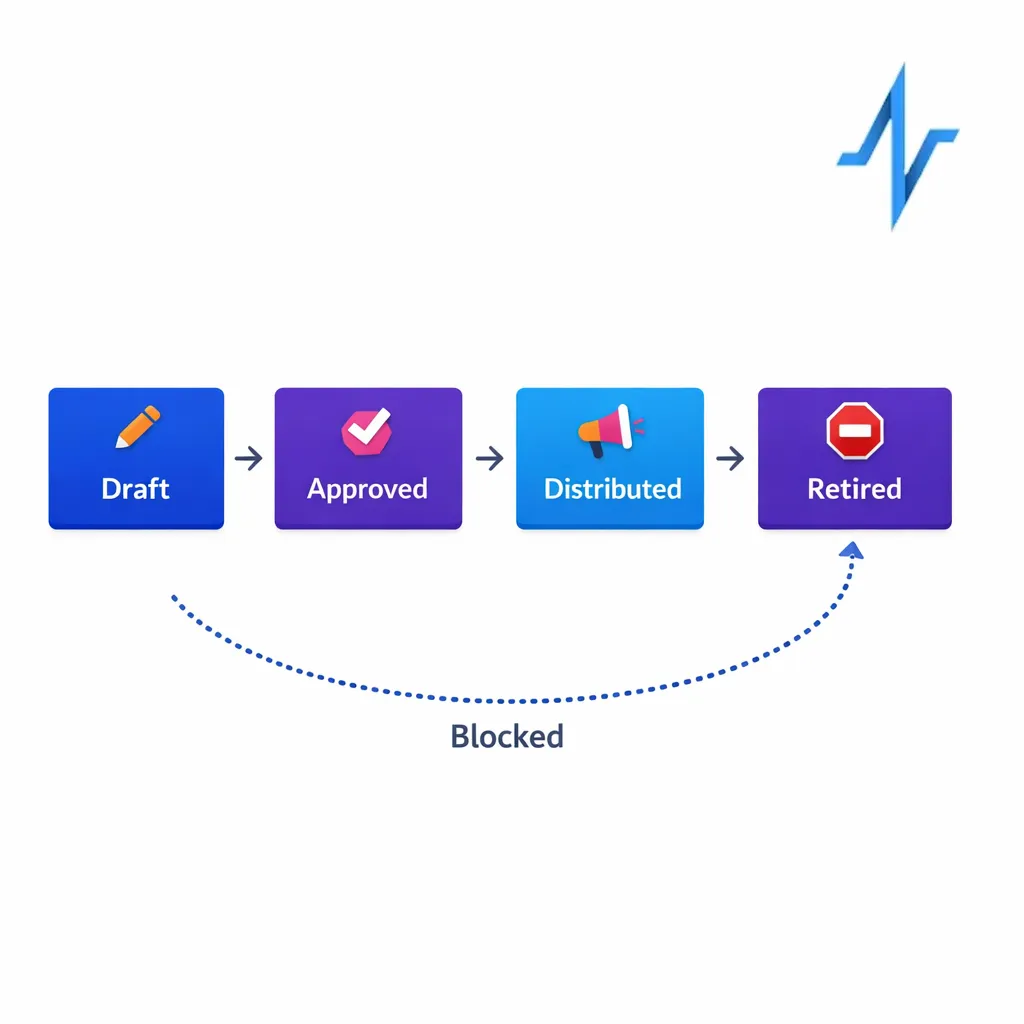
If your platform supports promotional content distribution, require strong audit trails and content-to-audience traceability. In the US, prescription drug promotion oversight sits with the FDA Office of Prescription Drug Promotion (OPDP), so your RFP should push vendors to show how they document what was delivered, to whom, when, and under which approved version.
RFP questions for content governance
- How do you manage content versioning and retirement, and how do you prevent retired assets from being used in journeys?
- What audit logs exist for content changes, targeting changes, and journey changes? How long are logs retained?
- How do you prove “what was shown” at the individual level while respecting privacy constraints and access controls?
3) Electronic records and signature considerations
If your workflows rely on electronic records or electronic signatures for regulated processes, clarify whether you need alignment with 21 CFR Part 11 expectations. Even when Part 11 is not strictly required for a marketing workflow, many teams borrow its discipline around audit trails, access controls, and record integrity to reduce operational risk.
Security requirements (ISO 27001, due diligence, and proof)

“Security requirements ISO 27001” often shows up in RFP templates, but the real question is whether the vendor can demonstrate control maturity and provide evidence. Ask for artifacts and processes, not just certifications, and require transparency about scope.
Baseline security and assurance requirements
- ISMS and certification scope: if claiming certification, specify whether it is aligned to ISO/IEC 27001:2022 and ask what products, locations, and teams are in scope.
- Attestation reports: request relevant SOC reports under NDA, including the period covered and any exceptions.
- Framework mapping: ask for a control mapping to a framework such as the NIST Cybersecurity Framework to simplify internal review.
- Access control: SSO, MFA, RBAC, least privilege, and partner access models.
- Logging and monitoring: audit logs, SIEM integration options, alerting, and retention.
- Encryption: encryption in transit and at rest, key management approach, and rotation policies.
- Resilience: backups, disaster recovery objectives, and restoration testing cadence.
RFP questions for vendor due diligence pharma teams actually need
- Provide your current ISO certification details (standard version, certification body, scope statement) and explain any scope exclusions.
- Provide the executive summary for your most recent SOC report and describe any noted exceptions and remediation status.
- Describe your vulnerability management program, including how you prioritize remediation and how customers are notified of critical issues.
- How do you separate customer data in a multi-tenant model, and what controls prevent cross-tenant access?
- What is your process for onboarding and offboarding agency users, and how quickly can access be removed?
Implementation and onboarding requirements (avoid “go-live and forget”)
Implementation and onboarding pharma teams succeed when the vendor is explicit about roles, effort, and what must be standardized. Your RFP should require a plan that includes change management, not just technical setup.
Implementation requirements
- Timeline with dependencies: including data readiness, integration lead times, and legal/security review milestones.
- Operating model: who owns journeys, data definitions, content tagging, and reporting definitions after launch.
- Enablement: training for brand teams, ops, agencies, and admins; certification paths; office hours.
- Migration plan: legacy journeys, lists, consent records, content libraries, and historical analytics where needed.
RFP questions for implementation
- Provide a sample project plan for a first brand launch and a second brand rollout. What gets faster and why?
- What are the top 10 reasons implementations slip, and what mitigations do you use?
- How do you support ongoing releases without breaking validated workflows and reporting definitions?
SLA, support, and release management requirements

Support and SLAs should be evaluated as operational risk controls, not procurement formalities. Your RFP should specify the level of support required during launches, major campaigns, and incident response.
SLA and support requirements to include
- Support hours and response targets: define P1/P2/P3 categories and your business-hour coverage needs.
- Escalation model: named roles, escalation path, and executive escalation for material incidents.
- Release communication: advance notice, change logs, and how breaking changes are handled.
- Performance expectations: page load and job execution expectations for high-volume sends and reporting.
RFP questions for SLAs
- Provide your standard SLA and your historical uptime reporting approach.
- How do you communicate platform incidents and post-incident RCA documentation?
- How do you handle urgent changes for regulated programs without compromising auditability?
Scoring framework: a practical way to compare vendors
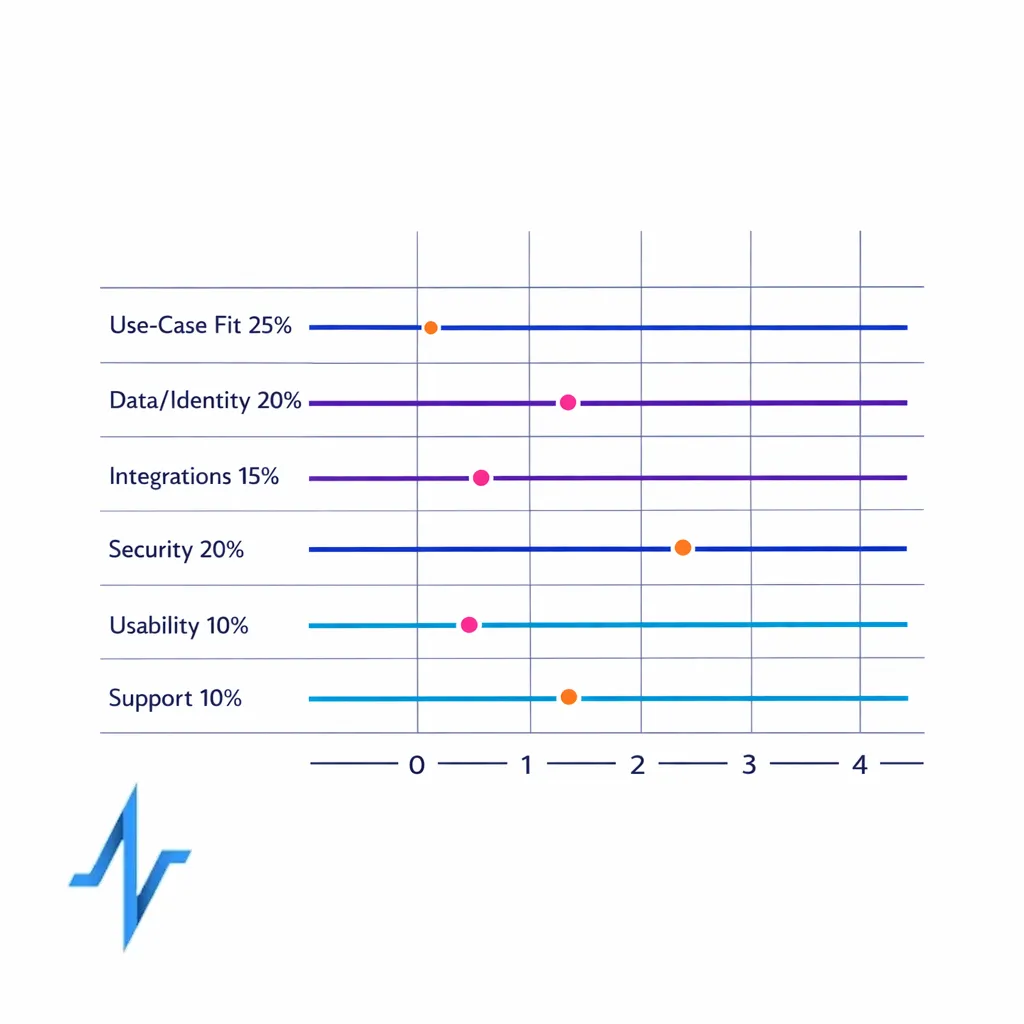
A scoring framework reduces politics and forces tradeoffs into the open. Use a weighted rubric, define minimum thresholds, and separate “capability fit” from “implementation risk.”
Recommended scoring categories (example weights)
- Use-case fit (25%): journey orchestration, HCP engagement workflows, patient enrollment, content operations.
- Data, identity, measurement (20%): first-party data support, explainable identity resolution, exports, metric governance.
- Integration and architecture (15%): CRM integration requirements, monitoring, environments, extensibility.
- Security and compliance (20%): ISO/SOC evidence, access controls, audit logs, incident readiness.
- Operational usability (10%): marketer self-serve, admin overhead, agency collaboration model.
- Implementation and support (10%): plan quality, enablement, SLAs, references.
Score each requirement on a 0–4 scale
- 0: Not available or not credible.
- 1: Available only with heavy custom work or unclear ownership.
- 2: Available with configuration and documented limitations.
- 3: Available, proven, and fits the operating model with minimal workarounds.
- 4: Available with strong evidence, plus accelerators (templates, governance tooling, analytics packages) that reduce time-to-value.
Minimum thresholds (avoid expensive surprises)
- Security: no vendor advances without evidence packages (certification scope and/or SOC summary, incident response process, access control model).
- Integration: no vendor advances without a clear integration architecture and operational monitoring plan.
- Measurement: no vendor advances without a concrete plan for metric definitions, exports, and data governance.
Common mistakes and misconceptions in pharma martech vendor evaluation
Most RFP failures are avoidable. They come from ambiguous scope, missing governance requirements, and confusing “has features” with “works in our operating model.”
Mistake 1: Treating identity and measurement as a “Phase 2” detail
If you do not define identity rules, consent enforcement expectations, and export requirements upfront, you will discover late that measurement is incomplete or not trusted. Put identity and measurement requirements in the core RFP, not the appendix.
Mistake 2: Over-indexing on channel checklists
Listing every channel can make vendors look similar and hides the real differentiators: orchestration logic, governance, usability, and integration resilience. Prioritize the workflows that drive speed and compliance, then test the vendor’s ability to execute those workflows in a demo.
Mistake 3: Accepting “ISO 27001” as a one-word answer
Even when a vendor is certified, the certification scope may not cover the product or operational teams you will use. Ask for scope statements, evidence, and how security controls are operated day-to-day, not just a logo on a slide.
Mistake 4: Ignoring the agency operating model
Agency partners often build journeys, manage content tagging, and run reporting. Your RFP should require a partner access model that supports collaboration without over-permissioning or breaking data governance.
What to do next (RFP-ready checklist)
- Write the one-paragraph scope: audiences, channels, systems in scope, and decisioning/measurement needs.
- Define 4–6 success metrics: speed, adoption, data quality, reporting effort reduction, measurable engagement lift.
- Choose 8–12 “demo scenarios”: real workflows your team will run (not generic feature tours).
- Lock your data governance basics: event taxonomy owner, data dictionary owner, metric definitions owner.
- Publish integration assumptions: CRM system(s), warehouse/BI tools, SSO, consent tools, monitoring requirements.
- Require security evidence: ISO scope, SOC summary under NDA, incident response process, access control model.
- Use the scoring rubric: weights, thresholds, and a single decision log shared across brand, ops, and IT.
- Plan the rollout model: pilot brand, second brand, and the operating model for scale.
Request a Demo, book a consultation, or see how Pulse Health supports the RFP requirements
If you are building an RFP for an engagement cloud and want to pressure-test requirements across orchestration, data governance, and analytics, Pulse Health can help you translate goals into an evaluation plan that brand, ops, and IT can all sign off on. You can Request a Demo or Book a Consultation to walk through your use cases, integration constraints, and what “good” measurement looks like for your team.
If you are already comparing vendors, ask for a platform walkthrough focused on real workflows (journey build, consent enforcement, audit trails, exports, and reporting). Bring your integration diagram and a short list of demo scenarios, and use the rubric above to keep the decision grounded in operational reality.